AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-11-12 View volume: 252
Nutritional endosymbiosis is widespread in nature and plays a central role in biological adaptation and evolution. Host cells harbor intracellular bacteria, forming a “metabolic factory” that relies on extensive metabolic exchange between host and endosymbiont. Compared to well-studied nutrient hijacking mechanisms in parasitic relationships, the nutrient transfer mechanisms in intracellular mutualistic symbiosis remain poorly understood. Although genomic and transcriptomic studies show expansion of host transporter gene families, endosymbiont genomes often retain very few transporter genes. How endosymbionts acquire nutrients from the host and exchange metabolites between bacteria remains an unresolved mystery.
This study uses the rice weevil (Sitophilus oryzae) and its endosymbiont Sodalis pierantonius as a model system. S. pierantonius provides the host with scarce vitamins, cofactors, and amino acids from grains—especially the aromatic amino acid phenylalanine (used for cuticle synthesis to enhance resistance to desiccation and biotic stress). The bacteria retain a complete shikimate pathway, generating phenylalanine from carbohydrate metabolism. Weevils lacking symbionts cannot develop on a pure starch diet, whereas symbiotic individuals develop normally, indicating that bacteria utilize carbohydrates for proliferation and supplement host nutrition. Bacterial cell transcriptomes show highly active glycolysis and polyol pathways, rich in carbohydrates. Combining high-pressure freezing (HPF), volume electron microscopy, and in situ high-resolution chemical analysis, this study reveals how endosymbionts efficiently acquire host carbohydrates.
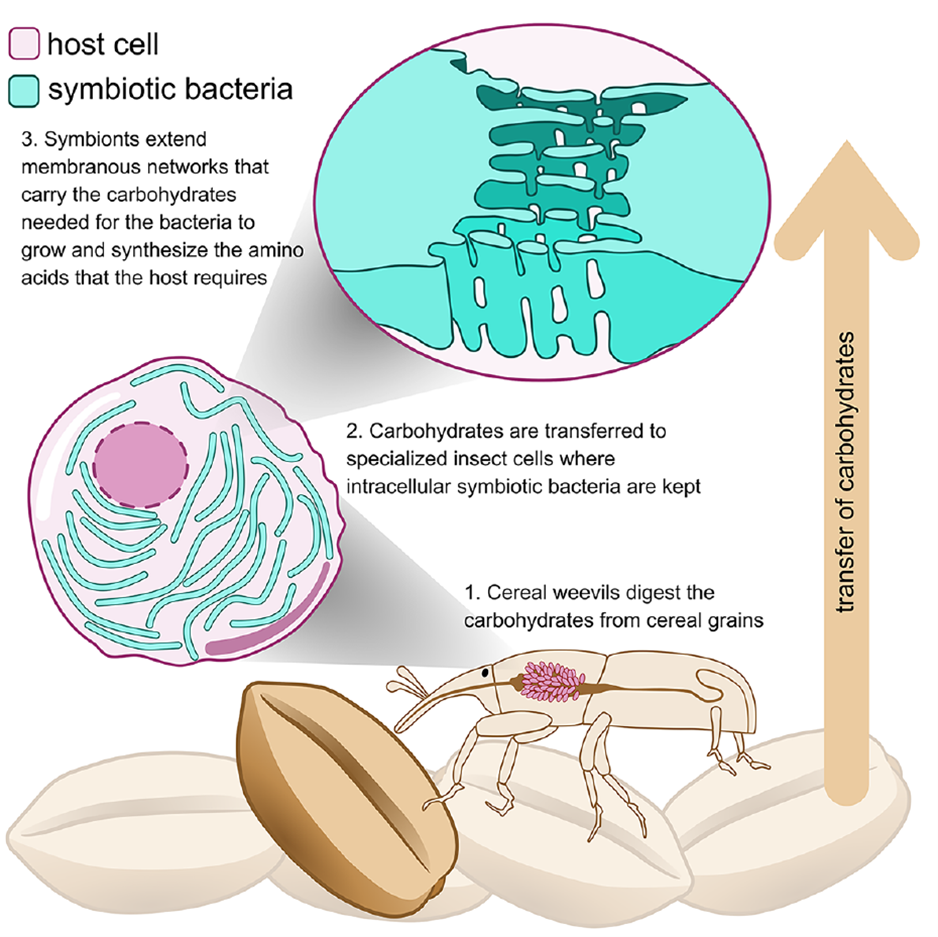
Figure 1. Graphical Abstract
Host Gut Epithelium Forms Extensive and Polarized Vesicle Networks
The study found that the bacteriome in rice weevil larvae has no direct contact with the gut lumen. To clarify how dietary nutrients reach the symbionts, high-pressure freezing combined with transmission electron microscopy revealed dense vesicle networks within gut epithelial cells. Immunofluorescence labeling with Rab5 and Rab7 showed Rab5 concentrated at the apical side (lumen-facing) and Rab7 toward the basal side (bacteriome-facing), forming a distinct polarized distribution. In aposymbiotic larvae, Rab5 polarity was reduced, indicating that endosymbionts enhance host vesicle polarization. This polarized pattern suggests an active transcytosis mechanism, enabling efficient trans-epithelial transport of carbohydrates and other nutrients from the gut lumen to the bacteriome, providing an upstream channel for endosymbiont nutrient acquisition.
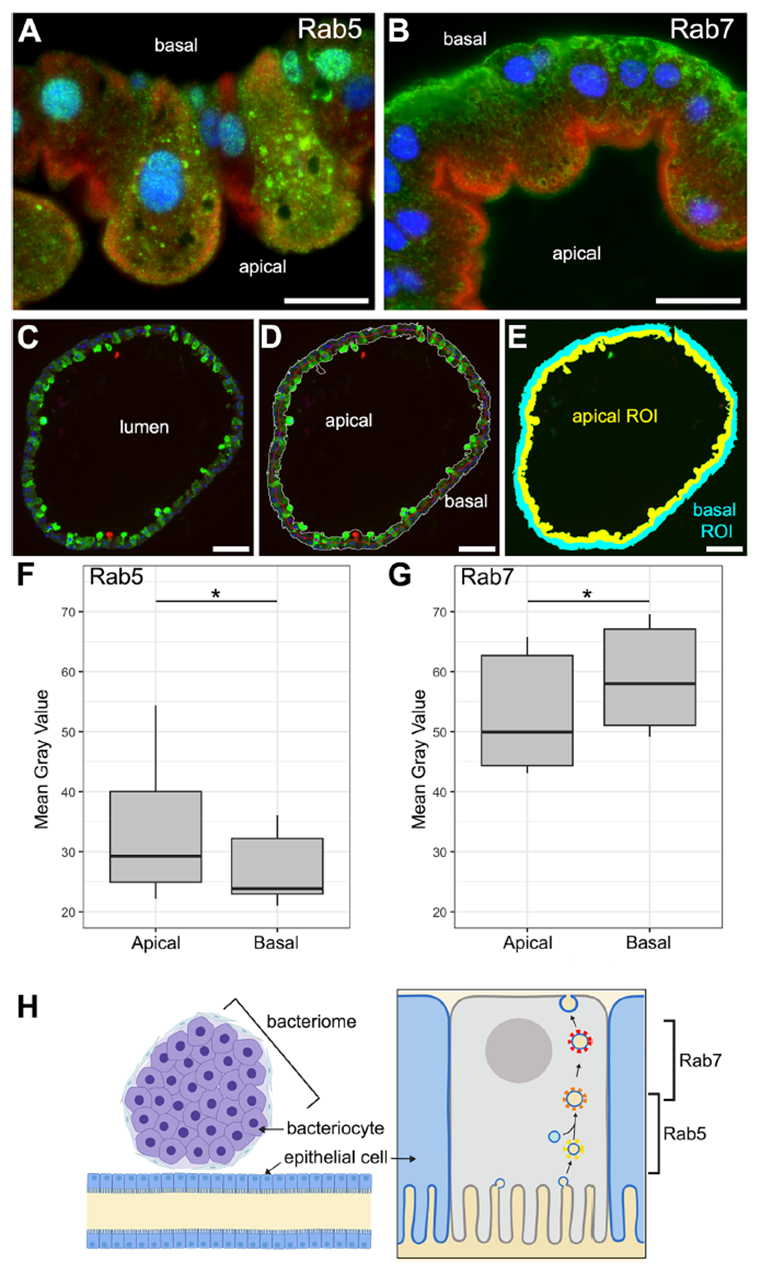
Figure 2. Host gut epithelium exhibits extensive and polarized vesicle networks
Endosymbionts Form Numerous Tubular Membrane Extensions
During cytoplasmic staining of bacterial cells, the study unexpectedly discovered abundant membrane material filling the space between endosymbionts, confirming that S. pierantonius resides directly in the host cytoplasm without a symbiosome membrane. Ultrastructural analysis revealed tubular structures ~20 nm in diameter and over 200 nm long, originating from the bacterial outer membrane and distributed between bacteria or between bacteria and vesicles. Immunogold labeling confirmed the presence of bacterium-specific outer membrane components, establishing bacterial origin. Electron tomography showed that tubules align longitudinally along bacteria, repeatedly anchor to the outer membrane, and interconnect to form complex three-dimensional networks—termed tubenets—far exceeding the isolated linear features of traditional nanotubes.
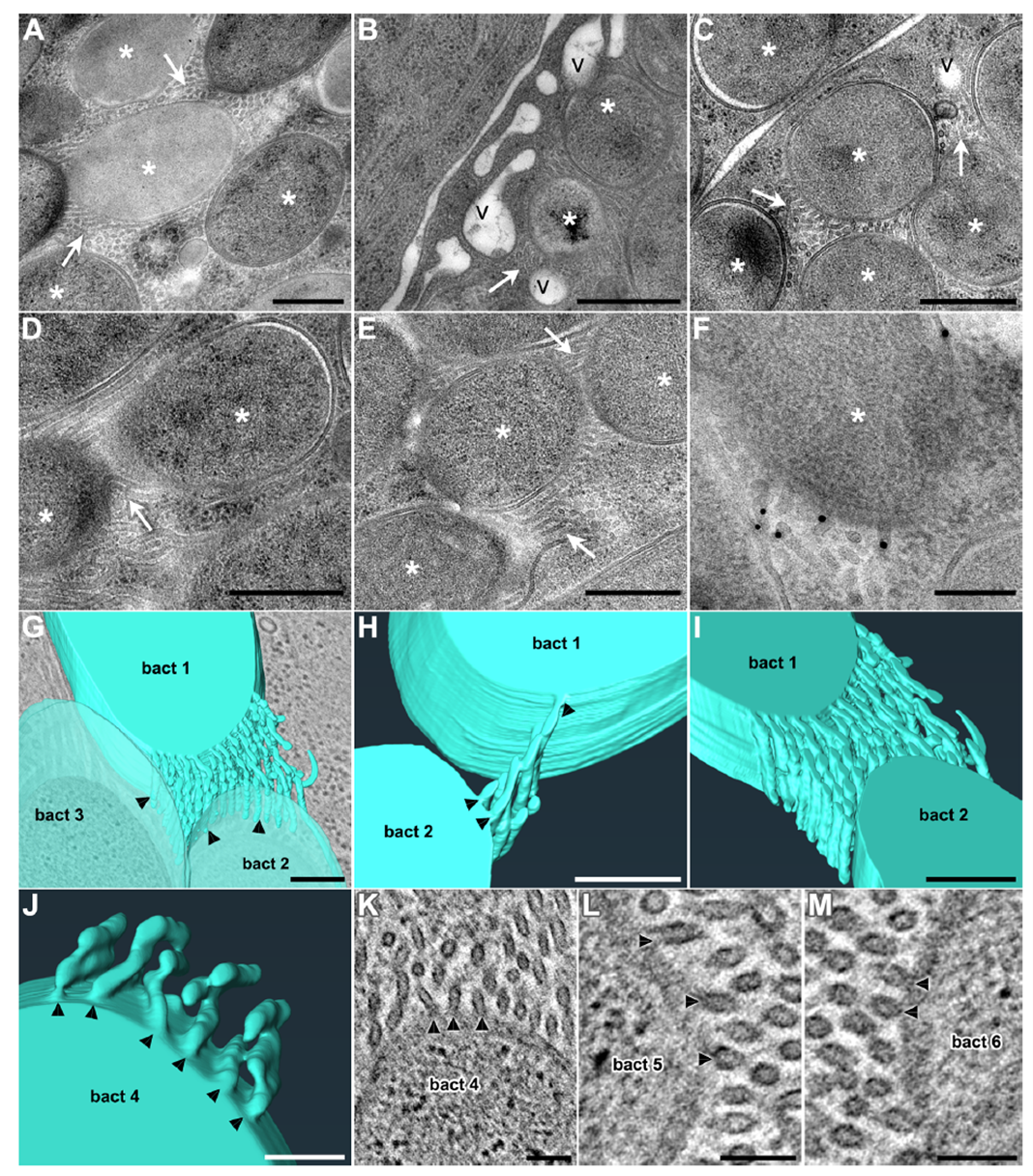
Figure 3. Endosymbionts generate abundant tubular membrane extensions inside bacterial cells
Tubenets are present throughout the larval, pupal, and adult life stages of S. oryzae and are widespread in S. zeamais with high symbiotic load, but absent in ancient endosymbiont hosts with highly reduced genomes (R. dominica, O. surinamensis) and low-load S. granarius. This distribution pattern indicates that tubenets are actively constructed membrane networks by S. pierantonius, evolving in sync with bacterial growth intensity and host nutritional demands, dramatically expanding the bacterium-host cytoplasmic interface and laying a structural foundation for nutrient exchange.
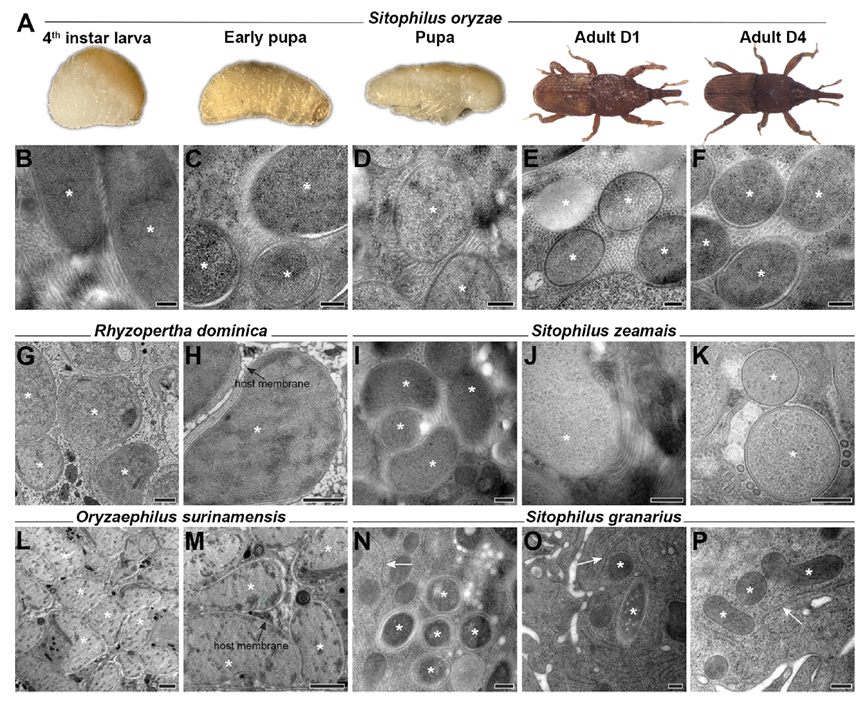
Figure 4. Prevalence of tubular structures across life stages of S. oryzae and other coleopteran species
Endosymbiont Tubenets and Host Vesicles Are Enriched with Carbohydrates
Young adult stages (when bacteria provide amino acids critical for cuticle synthesis) were selected for in situ carbon chemistry analysis using STXM. From four image stacks, 85 ROI carbon spectra were extracted, background-corrected, Z-score normalized, and clustered. Results showed highly similar carbon spectra between tubenets and host vesicles, distinctly separated from mitochondria, bacterial cytoplasm, and host cytoplasm. Correlation analysis with a standard spectral library further indicated a strong association of tubenets and vesicle spectra with carbohydrates, while other regions leaned toward proteins and amino acids. Pixel-level correlation mapping visually confirmed this enrichment pattern, establishing chemical equivalence in carbohydrate content between tubenets and vesicles.
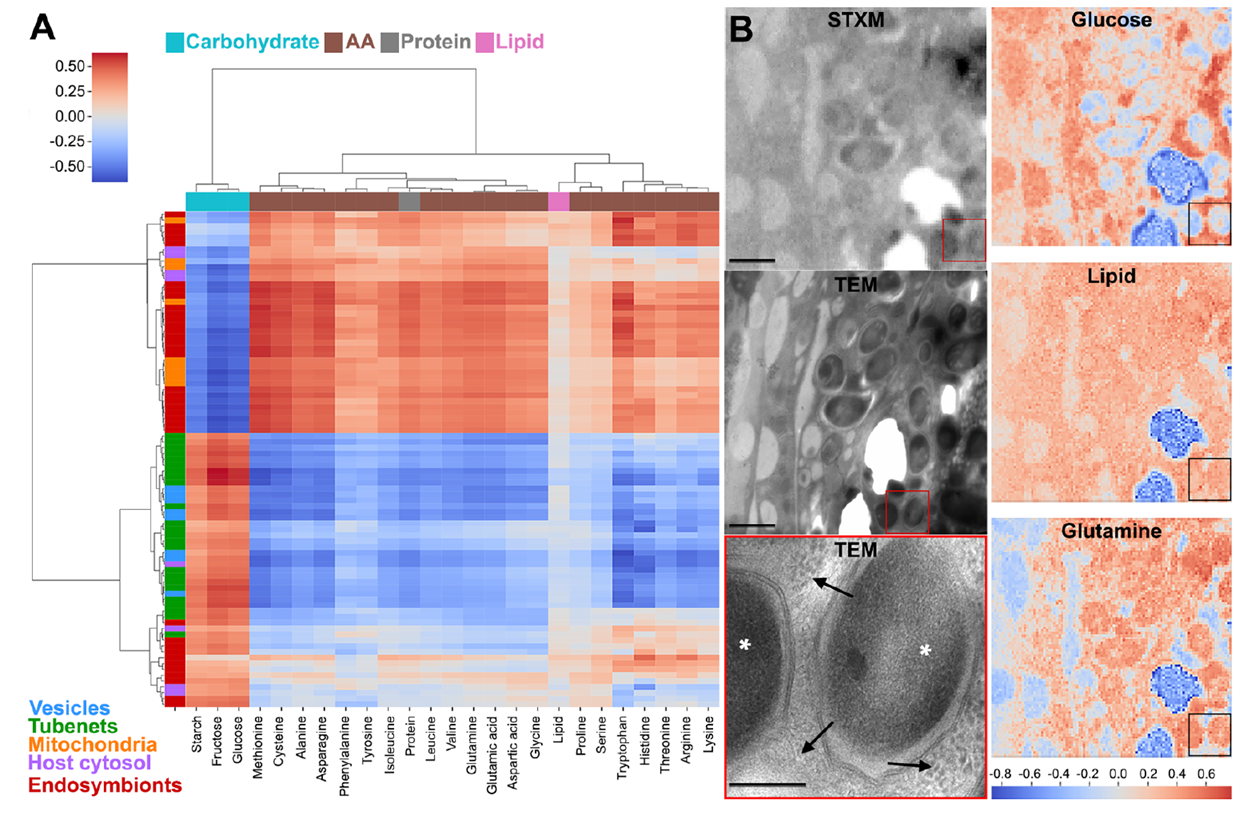
Figure 5. STEM analysis of carbon content in bacterial components
Reference spectrum modeling showed that tubenet spectra required higher carbohydrate coefficients for fitting, with statistical comparisons confirming no significant difference from vesicles but significantly higher than other ROIs. Gut HPLC revealed glucose as the dominant soluble sugar, and S. pierantonius highly expresses PTS glucose/N-acetylglucosamine transporters (ptsG, crr, nagE) and OmpC, suggesting passive diffusion of sugars via tubenets followed by active uptake. Integrated analysis indicates that tubenets and vesicles form a synergistic carbohydrate channel, enabling efficient uptake of metabolic substrates from the host cytoplasm by bacteria.
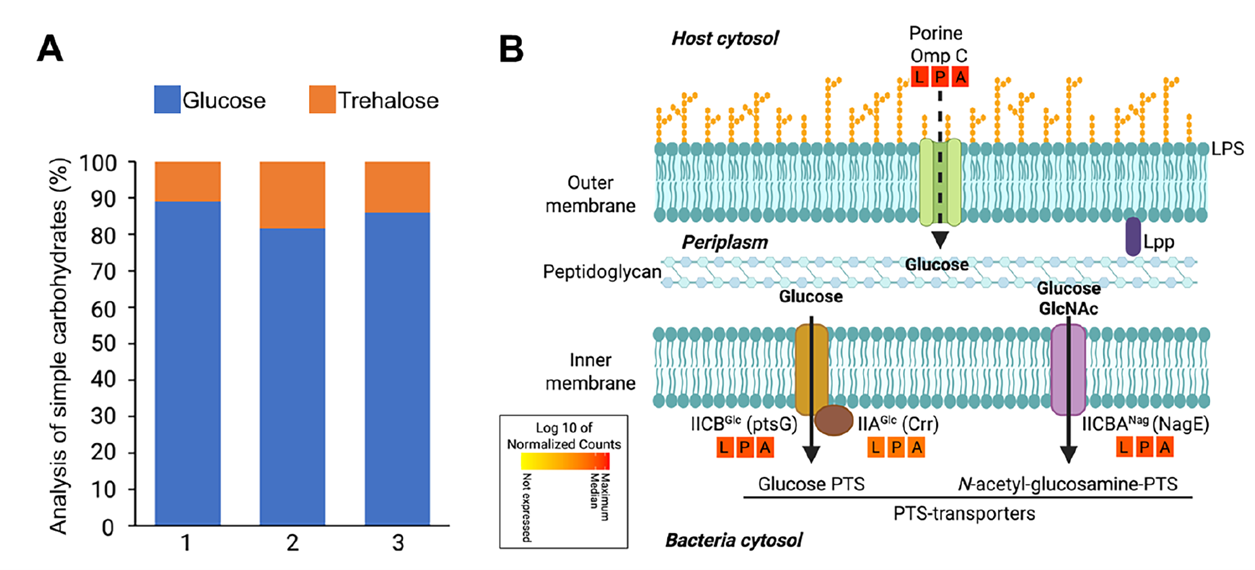
Figure 6. Major simple carbohydrates in the insect gut and glucose transporters in endosymbiont membranes
This study reveals that endosymbionts acquire nutrients—particularly carbohydrates—from host cells using complex membrane tubular networks (tubenets). These structures increase the bacterium-host cytoplasmic interface, potentially fuse with host vesicles, or facilitate inter-bacterial transfer, promoting carbohydrate acquisition to support bacterial growth and host amino acid production. Tubenets represent a bacterial-evolved “biological strategy” analogous to microvilli in multicellular intestines or root systems, enhancing nutrient absorption. This adaptation supports niche expansion in the Sitophilus/Sodalis association. The findings highlight that intracellular symbiosis relies on massive metabolic exchange and suggest possible convergent adaptations in other symbioses.
AtaGenix provided rabbit polyclonal antiserum against Braun’s lipoprotein (Lpp) (anti-Lpp antibody) for this study. This antibody was used in immunogold labeling experiments to specifically recognize the key protein Lpp that links bacterial peptidoglycan and outer membrane. Combined with lipid A and OmpC labeling, it conclusively proved that the tubular network (tubenets) originates from the S. pierantonius outer membrane, providing core protein evidence for its bacterial origin and membrane continuity.
As of November 11, 2025, over 400 publications have cited AtaGenix’s one-stop protein and antibody development technical services. Looking ahead, AtaGenix will continue to fully support scientific research, driving technological innovation and breakthroughs.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

