AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-11-05 View volume: 282
Duck Hepatitis A Virus Type 3 (DHAV-3) is a major pathogen responsible for duck viral hepatitis, posing a serious threat to the health of the meat duck industry. Although prior studies have shown that infection triggers a robust type I interferon (IFN-I) response, how the virus interacts with the host innate immune system across different infection stages remains unclear. Researchers hypothesized that key host immune molecules such as IFIH1, IRF7, and MAVS may perform differential antiviral roles depending on the infection phase. Using primary hepatocytes from Peking ducks as a model, researchers from the Institute of Animal Science, Chinese Academy of Agricultural Sciences, systematically analyzed expression changes in IFN-α, IFN-β, and eight related innate immune molecules following DHAV-3 infection. Through gene knockdown and overexpression experiments, they uncovered the regulatory patterns of these molecules at different infection stages, thereby elucidating the host’s dynamic immune mechanisms against DHAV-3.
Dynamic Changes and Functional Validation of IFN-I
The research team first examined the expression of type I interferons following DHAV-3 infection. Results showed that IFN-α transcription was transiently upregulated early in infection but subsequently strongly suppressed by the virus, whereas IFN-β levels continued to rise, peaking at 24 hours. Western blot analysis further confirmed that IFN-β protein levels were significantly higher than those of IFN-α, indicating a staged interferon response during infection. Detection of IFN-α and IFN-β proteins utilized high-specificity rabbit monoclonal antibodies provided by AtaGenix, ensuring accuracy at the protein level. Knockdown of IFN-I using small interfering RNA markedly increased viral replication, while overexpression of IFN-α effectively inhibited it, demonstrating that IFN-I, particularly IFN-α, plays a critical role in early defense against DHAV-3.
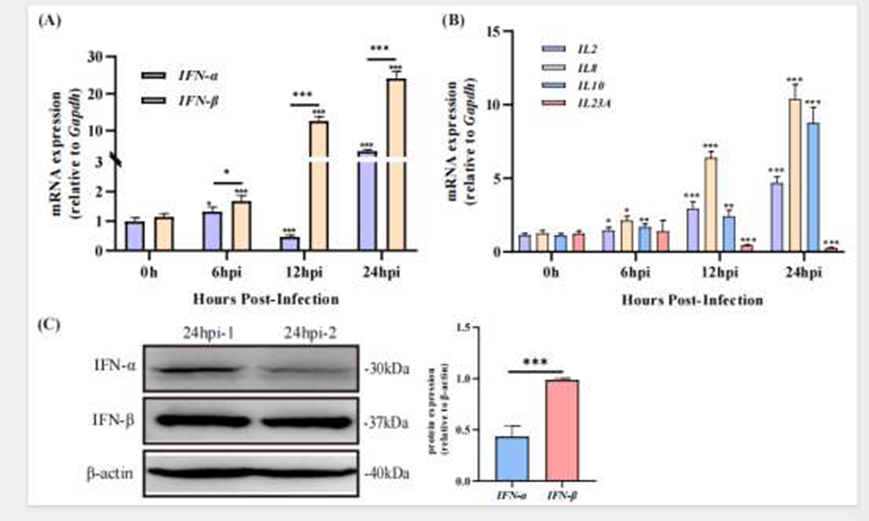
Figure 1. Changes in IFN-I and Cytokine Expression Following DHAV-3 Infection
Validation of Gene Knockdown and Overexpression Systems
Before investigating the functions of individual immune molecules, the researchers first validated the experimental system’s effectiveness. For eight key genes (IFIH1, IRF1, IRF7, MAVS, RIG-I, TBK1, TLR3, TLR4), they designed small interfering RNAs (siRNAs) and pcDNA3.1-based overexpression plasmids, which were transfected into primary duck hepatocytes. qRT-PCR validation confirmed that both the siRNA and plasmid systems achieved efficient and specific regulation, enabling stable gene silencing and overexpression in primary duck hepatocytes. This system provided a reliable technical foundation for subsequent studies on the functional differences of immune molecules during DHAV-3 infection and ensured the accuracy and reproducibility of viral replication and interferon regulation experiments.
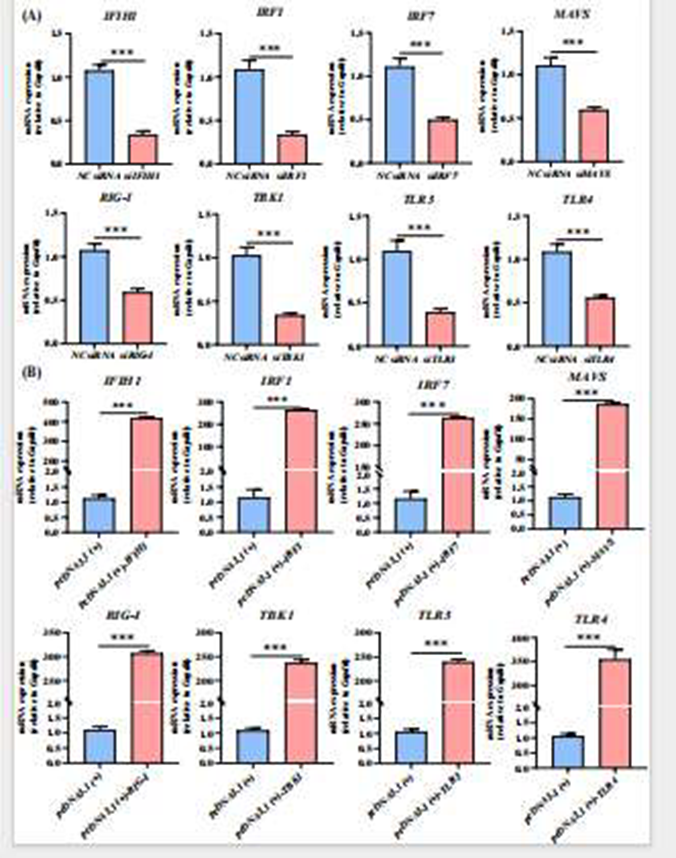
Figure 2. Gene Silencing and Overexpression Efficiency Measured by qRT-PCR
Regulatory Roles of Immune Molecules in DHAV-3 Infection
In the infection model, the researchers systematically analyzed the regulatory effects of eight host molecules closely linked to the type I interferon pathway (IFIH1, IRF1, IRF7, MAVS, RIG-I, TBK1, TLR3, and TLR4) across different stages of DHAV-3 infection. Through gene knockdown and overexpression experiments, they revealed that the roles of these signaling molecules in viral replication and interferon response were not static but exhibited clear stage-specific differences as infection progressed.
In the early stage of infection, IRF7 and TLR4 were found to play critical roles in the host’s initial defense. Knockdown of IRF7 or TLR4 significantly increased viral replication, indicating their essential role in early suppression of DHAV-3. In contrast, TLR3 knockdown reduced viral replication, suggesting it may be exploited by the virus early on. Meanwhile, overexpression of IFIH1 and MAVS effectively restricted viral proliferation and was accompanied by increased IFN-α and IFN-β expression, with IFIH1 primarily enhancing IFN-α signaling and MAVS activating both interferon types. Collectively, the IFIH1–MAVS–IRF7 axis forms the core antiviral signaling pathway in early infection, rapidly inducing interferon secretion to suppress viral replication.
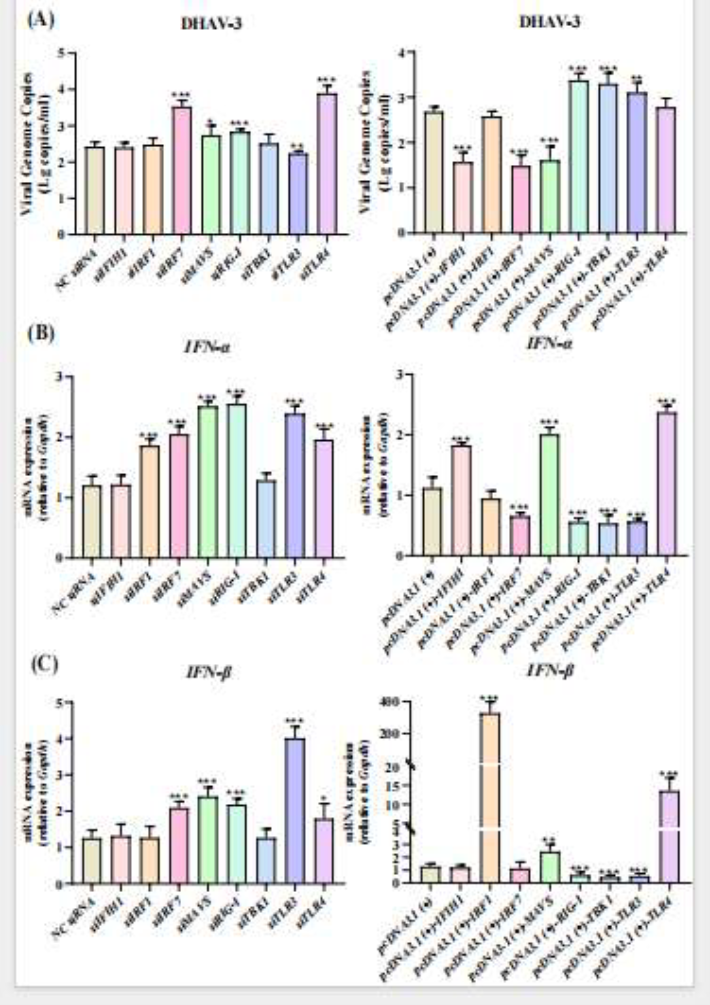
Figure 3. Effects of Gene Silencing and Overexpression on DHAV-3 Replication and IFN-I Response at 6 Hours Post-Infection
In the mid-stage of infection, a shift occurred in the immune interplay between host and virus. At this stage, knockdown of IRF7 and TLR4 unexpectedly reduced viral replication, while overexpression of MAVS and IRF1 surprisingly promoted viral proliferation, despite both molecules upregulating IFN-α and IFN-β expression. This suggests that during mid-replication, DHAV-3 may “hijack” the host interferon system via the MAVS pathway, triggering inflammation at the cost of its own replication. In contrast, overexpression of IFIH1 and TLR3 maintained strong antiviral effects, indicating greater signal stability and resistance to viral counter-exploitation.
Figure 4. Effects of Gene Silencing and Overexpression on DHAV-3 Replication and IFN-I Response at 12 Hours Post-Infection
In the late stage of infection, regulatory trends across multiple signaling pathways shifted again. Knockdown of IRF7, MAVS, RIG-I, TBK1, TLR3, and TLR4 all significantly enhanced viral replication, while their overexpression generally suppressed it, indicating these molecules regain antiviral function late in infection. Notably, IFIH1 consistently inhibited DHAV-3 throughout the infection cycle and sustained IFN-α expression; overexpression of MAVS and IRF7 also markedly increased IFN-β levels, suggesting reactivation of classic interferon antiviral mechanisms in late infection. Overall, IFIH1 acts as a stable antiviral “executor,” while IRF7 and MAVS switch roles across stages—suppressing virus early and late, but potentially exploited briefly in mid-infection. Together, they form a dynamically regulated antiviral network, balancing immune defense with prevention of immune damage.

Figure 5. Effects of Gene Silencing and Overexpression on DHAV-3 Replication and IFN-I Response at Later Stages
Relationship Between Type I Interferon and Viral Replication
The study further revealed a clear negative correlation between type I interferon (IFN-I) expression dynamics and DHAV-3 replication levels. Overall, when IFN-α and IFN-β expression was induced, viral replication was effectively suppressed; when interferon signaling was blocked or overactivated, the virus gained a replication advantage. Particularly in mid-infection, overexpression of MAVS and IRF1 significantly elevated interferon levels yet increased viral replication, suggesting excessive IFN signaling may be co-opted by the virus to disrupt immune balance. Moreover, different immune molecules regulated the two interferons inconsistently—IFIH1 primarily promoted IFN-α expression, IRF7 favored IFN-β upregulation, and TLR-family molecules exhibited bidirectional regulation depending on the stage. These differences collectively demonstrate that the host does not rely on sustained interferon release to combat the virus but instead maintains a dynamic balance between antiviral defense and avoidance of immune overreaction through precise signal modulation, achieving both tissue homeostasis and infection control.

Figure 6. Effects of Gene Silencing and Overexpression on DHAV-3 Replication and IFN-I Response
This study systematically elucidates the “stage-specific” antiviral roles of IFIH1, IRF7, and MAVS during DHAV-3 infection: IFIH1 consistently suppresses the virus throughout the cycle as a stable antiviral effector; IRF7 and MAVS exhibit functional shifts—dominating antiviral responses early, potentially exploited by the virus in mid-infection, and regaining antiviral efficacy late. The research underscores that the host’s innate immune response is not a singular defense mechanism but a dynamic balancing system, with outcomes depending on infection stage and molecular synergy. These findings provide critical insights into the immune evasion mechanisms of duck hepatitis virus and inform related prevention and control strategies.
The high-specificity rabbit monoclonal antibodies for IFN-α and IFN-β used in this study were developed using AtaGenix’s proprietary Xten™ Mab single B cell monoclonal antibody platform. This platform, based on single B cell screening technology, offers high specificity, stability, affinity, and diversity, enabling rapid generation of functional antibodies against multiple species and targets—widely applied in pathogen research, signaling pathway analysis, and novel vaccine development.
More Technical Services and Collaboration Inquiries
Contact us at: info@atagenix.com or call 027-87001869 for tailored service solutions.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

