AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-20 View volume: 666
Project Snapshot — A cancer metabolism team investigating how SLK alternative splicing drives glycolysis needed a phosphorylation-specific antibody targeting the ENO1 Ser2 site. AtaGenix delivered a custom phospho-ENO1(S2) polyclonal antibody validated for WB, IP, and IHC, plus recombinant ENO1 wild-type and mutant proteins for functional controls — enabling a publication in Cancer Research (2025).
Abnormal energy metabolism is a hallmark of tumor cells, with the glycolysis pathway playing a particularly critical role. This study focused on an alternative splicing event of Ste20-like kinase (SLK), revealing that its variant SLKv enhances phosphoenolpyruvate (PEP) production by phosphorylating Enolase 1 (ENO1) at the Ser2 site, thereby accelerating glycolysis and driving tumor progression. Using liver cancer patient samples, cell lines, and animal models, the research team systematically elucidated the mechanism of the “SLKv–ENO1–PEP” metabolic acceleration axis. The findings were published in Cancer Research (2025, DOI: 10.1158/0008-5472.CAN-25-0523).
The research team required: (1) a highly specific phosphorylation antibody targeting the ENO1 Ser2 site that could distinguish phosphorylated ENO1 from the unmodified form across WB, IP, and IHC platforms; (2) recombinant ENO1 proteins — wild-type and phospho-mimetic/dead mutants (S2D/S2A) — as functional controls for mechanistic validation; and (3) supporting capabilities including phage display screening and multi-platform functional validation to confirm the antibody’s specificity and the biological significance of Ser2 phosphorylation.
Developing a phospho-specific antibody against ENO1 Ser2 presented multiple difficulties:
AtaGenix designed an integrated workflow combining phospho-antibody development and recombinant protein production:
The custom phospho-ENO1(S2) antibody enabled the research team to accurately confirm the phosphorylation status of ENO1 at key nodes of the glycolysis pathway. Combined with the S2A/S2D mutant proteins, the team obtained reproducible molecular evidence that SLKv-mediated Ser2 phosphorylation enhances ENO1 catalytic activity, increases PEP production, and accelerates glycolysis in hepatocellular carcinoma. The study further demonstrated that targeting SLKv with antisense oligonucleotides effectively inhibits glycolysis and tumor growth, establishing the SLKv–ENO1–PEP axis as a promising metabolic target for cancer therapy.
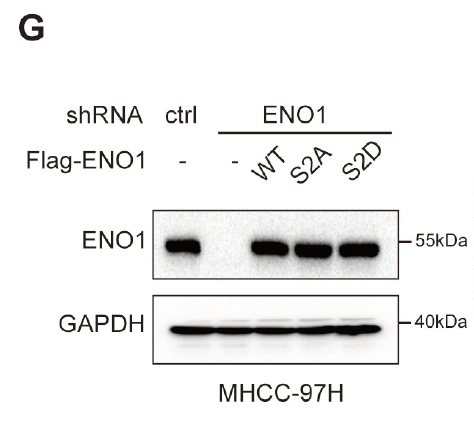
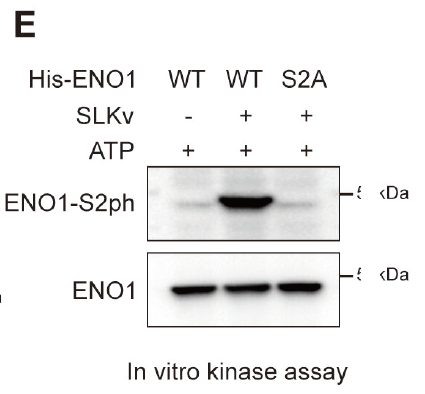
Figure 1. Validation of ENO1 Ser2 phosphorylation using AtaGenix custom phospho-specific antibody. Left: phosphorylation detection in tumor cell lysates. Right: functional analysis of SLKv-mediated ENO1 phosphorylation in glycolysis pathway regulation.
About AtaGenix
AtaGenix provides one-stop customized support from antibody/protein design to functional validation, covering custom antibodies (including phospho-specific), protein expression across five systems, phage display screening, and multi-platform validation. Learn more at www.atagenix.com.
This case study is based on a published research collaboration. Results may vary depending on target antigen, modification type, and experimental conditions. All proprietary client information is subject to NDA. Reference: Cancer Research. 2025. DOI: 10.1158/0008-5472.CAN-25-0523
Need a phosphorylation-specific antibody for your signaling or metabolism research? AtaGenix delivers from phospho-peptide design through two-round purification to multi-platform validated antibody.
Explore Anti-PTM Antibody ServicePeptide Synthesis | E. coli Expression | Polyclonal Antibody | Phage Display
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

