AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-04 View volume: 468
Project Snapshot — A sericulture virology team studying how BmNPV reprograms host amino acid metabolism needed a highly specific ATG8 polyclonal antibody to detect autophagy markers in silkworm cells. AtaGenix delivered a custom rabbit anti-ATG8 antibody with ELISA titer >1:64,000 and >90% purity, validated for IF and WB — enabling a publication in PLOS Pathogens (2025).
Bombyx mori nucleopolyhedrovirus (BmNPV) is a major pathogen in sericulture, causing substantial economic losses. The virus supports its replication by reprogramming host amino acid metabolism — specifically, BmNPV upregulates the amino acid transporter Slc7a6 to enhance arginine uptake and induces mitochondrial autophagy (mitophagy) to maintain intracellular amino acid levels, thereby fueling viral proliferation through an “exogenous uptake–endogenous supply” dual mechanism. The research was published in PLOS Pathogens (2025, DOI: 10.1371/journal.ppat.1013331).
The research team aimed to elucidate how BmNPV reprograms amino acid metabolism in silkworm cells. They required: (1) a highly specific ATG8 antibody to detect autophagy markers induced by BmNPV infection via immunofluorescence (IF) and Western Blot (WB); (2) compatibility with laser confocal microscopy for quantitative mitophagy analysis using Fiji software; (3) sufficient sensitivity to distinguish autophagy levels between BmNPV-infected and uninfected BmN cells; and (4) reliable performance alongside Slc7a6 knockout validation and viral titer (TCID50) assays.
Developing an ATG8 antibody for insect virology research presented specific hurdles:
AtaGenix designed a targeted antibody development workflow for this insect biology application:
The custom ATG8 antibody enabled the research team to validate that BmNPV infection induces mitochondrial autophagy in silkworm cells and that the virus maintains amino acid homeostasis through this autophagy-driven endogenous supply mechanism. Combined with Slc7a6 knockout experiments (demonstrating reduced arginine uptake and viral titer), qPCR targeting the viral gene vp39, and TCID50 viral titer assays, the team fully elucidated the “exogenous uptake (Slc7a6) – endogenous supply (mitophagy)” dual mechanism by which BmNPV reprograms arginine metabolism to support replication. These findings provide new molecular targets for developing antiviral strategies in sericulture, with significant agricultural and economic implications.
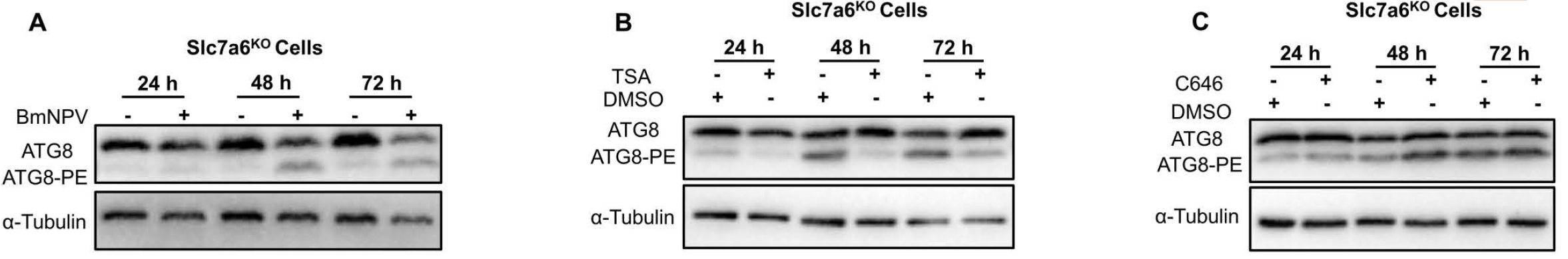
Figure 1. BmNPV-induced mitochondrial autophagy in silkworm cells. AtaGenix custom anti-ATG8 antibody enabled confocal IF detection of autophagy puncta and WB quantification of ATG8 lipidation, confirming the virus-driven mitophagy mechanism that maintains amino acid homeostasis for viral replication.
About AtaGenix
AtaGenix provides one-stop customized support from antibody/protein design to functional validation, covering custom polyclonal and monoclonal antibodies, protein expression, phage display, and multi-platform validation — including non-standard species (insect, plant, aquatic) where commercial antibodies are unavailable. Learn more at www.atagenix.com.
This case study is based on a published research collaboration. Results may vary depending on target protein, host species, and experimental conditions. All proprietary client information is subject to NDA. Reference: PLOS Pathogens. 2025. DOI: 10.1371/journal.ppat.1013331
Need custom antibodies for insect, plant, or non-model organism targets where commercial reagents don’t exist? AtaGenix designs species-specific immunogens and delivers validated antibodies for any host.
Explore Polyclonal Antibody ServiceRapid Hybridoma | Peptide Synthesis | E. coli Expression | Insect / BEVS
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

