AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-11-04 View volume: 937
Project Snapshot — A diagnostics company needed assay-grade SARS-CoV-2 spike protein with native glycosylation and conformational integrity. AtaGenix delivered purified antigen from gene sequence in 18 days, enabling the client to begin ELISA kit validation ahead of schedule.
The SARS-CoV-2 spike protein is a 1,273-amino-acid homotrimer with 22 N-linked glycosylation sites per monomer. Its receptor-binding domain (RBD) must fold correctly to present the ACE2-binding epitope that most diagnostic antibodies target. The client had tested an E. coli-expressed RBD fragment, but it lacked glycosylation, showed poor ELISA signal-to-noise ratios, and failed to capture patient serum antibodies consistently. They needed a full-length, properly folded spike protein — fast.
AtaGenix deployed its HEK293 transient expression platform with a codon-optimized spike construct containing a C-terminal His-tag and a furin cleavage site mutation (to stabilize the prefusion conformation). Key steps included:
The SDS-PAGE gel below shows the full purification workflow. Strong, clean bands in elution fractions E1–E8 confirm efficient target capture with minimal co-purifying contaminants:
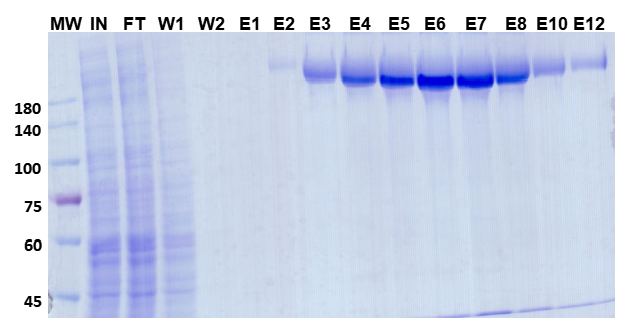
Figure 1. SDS-PAGE analysis of SARS-CoV-2 spike protein purification. MW = marker; IN = input; FT = flow-through; W1–W2 = wash fractions; E1–E12 = elution fractions. Strongest bands appear in E1–E8, confirming high-yield recovery with clean background.
The purified spike protein showed >95% trimer content by analytical SEC, with endotoxin below 0.1 EU/µg. When benchmarked against the client's previous E. coli-derived RBD, the mammalian-expressed full-length spike achieved a 3.5-fold improvement in ELISA signal-to-noise ratio for patient serum IgG detection. The client proceeded to validate their serological assay kit using AtaGenix-supplied antigen as the capture reagent.
Why This Matters
For viral antigens that rely on conformational epitopes and glycosylation, the expression system directly determines downstream assay performance. This project demonstrated that investing in mammalian expression upfront can eliminate months of troubleshooting caused by structurally compromised E. coli-derived antigens — particularly for serological diagnostics where sensitivity and specificity are non-negotiable.
This case study represents an anonymized project outcome. Results may vary depending on target protein, construct design, and project scope. All client information is subject to NDA. Specific timelines, yields, and QC specifications should be confirmed during project consultation.
Response within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

