AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2026-03-20 View volume: 379
Over the past 30+ years, therapeutic antibodies have evolved from traditional IgG monoclonal antibodies to a diverse array of derivatives: antibody–drug conjugates (ADCs), antibody–oligonucleotide conjugates (ARCs), bispecific/multispecific antibodies (BsAbs/MsAbs), immunocytokines, antibody fragments, and non-Ig scaffold proteins. These approaches improve solid tumor penetration, reduce off-target toxicity, and enable precise modulation of the tumor microenvironment — forming the translational backbone of modern cancer immunotherapy.
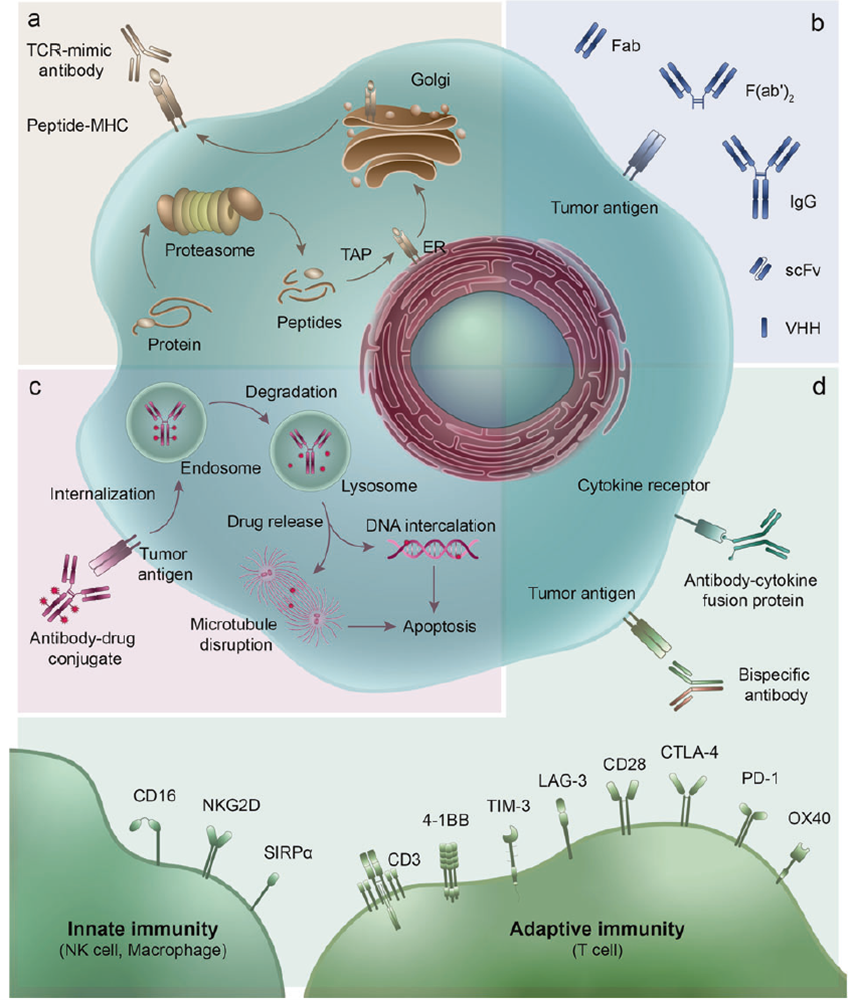
Figure 1. Representative therapeutic antibodies and their derivatives.
ADCs consist of a monoclonal antibody, a cytotoxic payload, and a linker, enabling targeted delivery via antigen-mediated receptor internalization. The FDA has approved 15 ADCs to date, including gemtuzumab ozogamicin (CD33, AML), brentuximab vedotin (CD30, HL/sALCL), trastuzumab deruxtecan (HER2+ breast cancer), sacituzumab govitecan (Trop-2, TNBC), and telisotuzumab vedotin (c-Met, NSCLC). Over 100 ADCs are in clinical development, with major focus on improving DAR homogeneity, site-specific conjugation, and cleavable linker design.
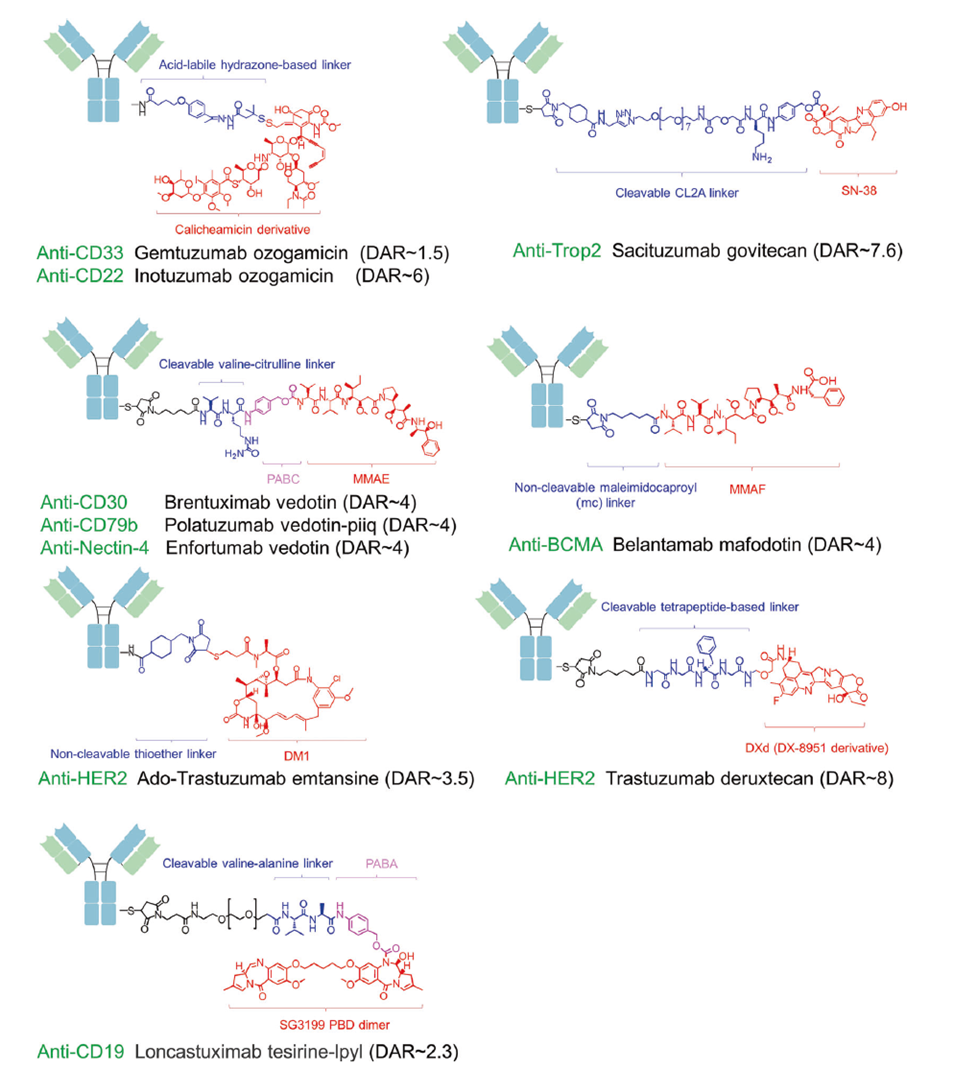
Figure 2. Schematic of FDA-approved antibody–drug conjugates (ADCs).
ARCs use antibodies as delivery vehicles to overcome the liver- and eye-restricted delivery limitations of siRNA and ASO therapeutics. Platforms such as Genentech's THIOMAB, Avidity's AOC, and Dyne's FORCE have advanced into clinical trials for muscular disorders and oncology. Key challenges remain in endosomal escape efficiency and quantitative analytics.
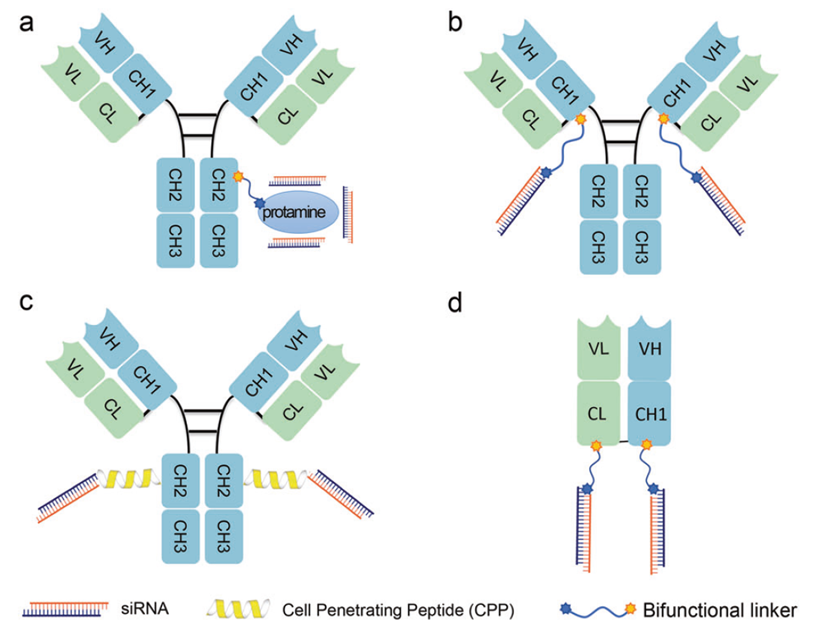
Figure 3. Schematic of antibody–siRNA conjugates (ARCs).
These molecules simultaneously engage two or more antigens for synergistic effects. Primary mechanisms include T-cell/NK-cell redirection (blinatumomab [CD19×CD3], tebentafusp [gp100/HLA-A2]), checkpoint blockade (PD-1×CTLA-4, PD-L1×4-1BB), and dual-pathway blockade (amivantamab [EGFR×MET], approved for NSCLC exon 20 insertion). Formats include BiTE, CrossMab, and Knobs-into-Holes. Hundreds of clinical trials are ongoing, with substantial progress in hematologic malignancies; solid tumors continue to face immunosuppressive TME barriers.
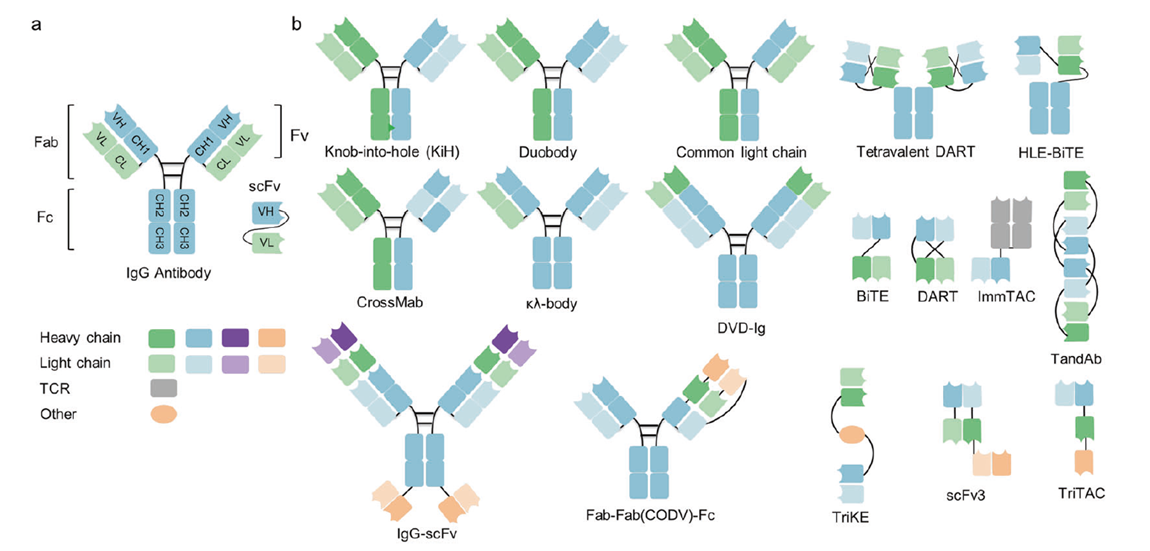
Figure 4. IgG structure and representative multispecific antibody formats in clinical development.
These fuse cytokines (IL-2, IL-12, TNF-α, GM-CSF) to antibodies to achieve high local concentrations in the tumor while minimizing systemic toxicity. Notable examples include L19-IL2, NHS-IL12, and bintrafusp alfa (PD-L1 + TGF-β trap). Clinical data demonstrate effective reversal of immune suppression in "cold" tumors, with several candidates advancing to phase III.
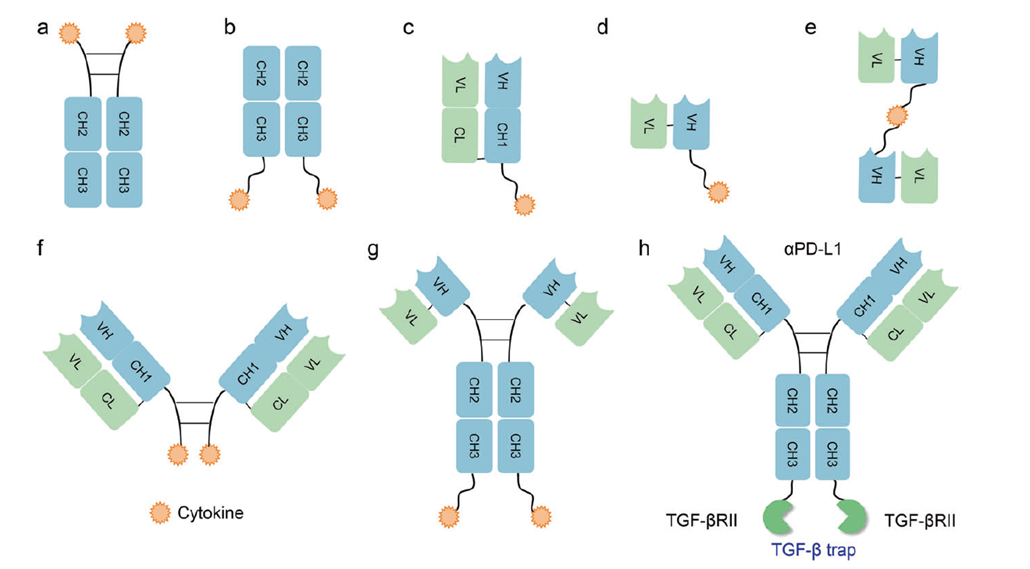
Figure 5. Different formats of antibody or antibody-fragment cytokine fusion proteins.
Smaller formats — Fab (~55 kDa), F(ab')₂, scFv (~25 kDa), and VHH nanobodies (~15 kDa) — markedly improve solid tumor penetration and avoid Fc-related toxicities. VHH nanobodies offer high stability, extreme pH/temperature tolerance, and long CDR3 loops accessing cryptic epitopes. Caplacizumab (VHH-based) is FDA-approved, and anti-HER2 VHH PET imaging agents are in phase II. Non-Ig scaffolds (DARPins, monobodies) further expand affinity range and manufacturability.
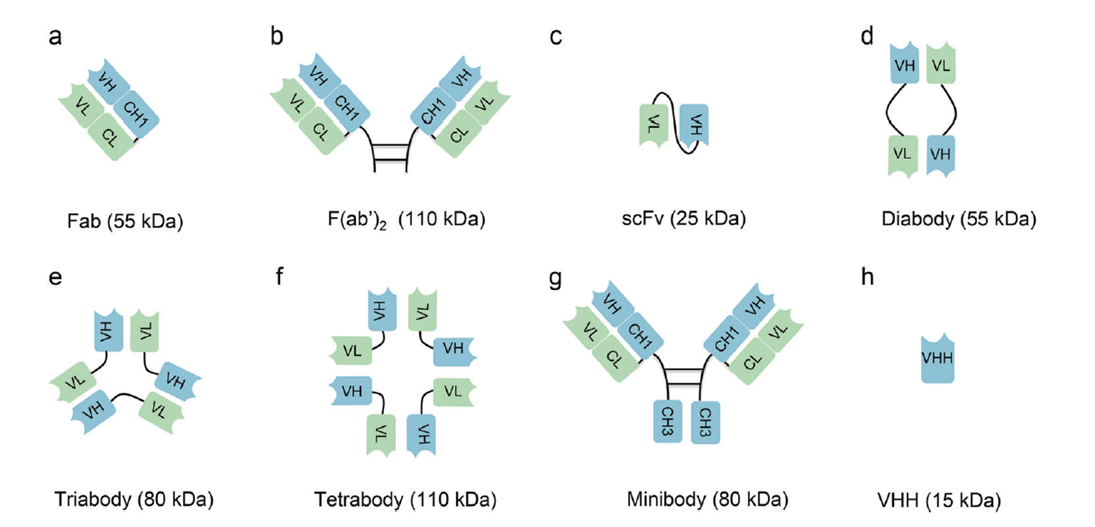
Figure 6. Different formats of antibody fragments and derivatives.
Novel formats continue to emerge, with revived interest in non-IgG immunoglobulins (IgA, IgM). Advances in site-specific conjugation, Fc-enhancing mutations, single-cell omics, and spatial transcriptomics are expanding the therapeutic reach of engineered antibody derivatives. Solid tumor challenges — insufficient T-cell infiltration and immunosuppressive TME — will likely require combination strategies pairing multispecific antibodies with checkpoint inhibitors, neoantigen vaccines, or oncolytic viruses.
Meanwhile, soluble TCRs, TCR-mimic antibodies (TCRm), and their derivatives (e.g., ImmTACs) enable targeting of HLA-restricted peptide neoantigens from "undruggable" intracellular targets (p53, KRAS). Integrated with next-generation sequencing and advanced bioinformatics, these approaches are ushering in a new era of antibody-mediated intracellular antigen targeting and precision oncology.
AtaGenix supports the full spectrum of therapeutic antibody engineering — from bispecific/multispecific development and ADC antibody production to VHH nanobody discovery and recombinant antibody expression. Share your project requirements and we'll design a customized development plan.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

