AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2026-04-03 View volume: 298
Since monoclonal antibodies (mAbs) ushered in a new era of cancer immunotherapy in the late 20th century, they have become a mainstay of clinical treatment. However, challenges such as tumor antigen heterogeneity, immune escape, and drug resistance have driven the rapid development of next-generation multispecific antibodies. Among them, bispecific antibodies (BsAbs) can simultaneously bind two different antigens, while trispecific antibodies (TsAbs) extend this capability to three antigen targets, enabling stronger immune redirection, signal blockade, and microenvironment regulation.
Bispecific antibodies (BsAbs) and trispecific antibodies (TsAbs) represent a major breakthrough in antibody engineering technology. They can simultaneously engage two or three different antigens or epitopes, thereby achieving precise immune cell redirection, dual-target blockade, or synergistic signal amplification. From its conceptual proposal in the 1960s to today, this technology has resulted in the approval of more than 17 anti-tumor BsAbs, with TsAbs now advancing into clinical trials. This marks a paradigm shift from monoclonal antibodies (mAbs) to multispecific platforms. Their core advantage lies in overcoming the limitations of tumor heterogeneity, antigen escape, and the immunosuppressive tumor microenvironment (TME), providing more effective and safer “off-the-shelf” solutions for cancer immunotherapy.
The core of bispecific and trispecific antibodies lies in the construction of an immune synapse. This process redirects effector cells (such as T cells) to tumor cells independently of MHC restriction, inducing perforin/granzyme-mediated lysis, cytokine release (IFN-γ, TNF-α, IL-2), and tumor cell apoptosis.
Common mechanisms of BsAbs: T-cell redirection (e.g., CD3 × TAA), dual signaling pathway blockade (e.g., PD-1 × VEGF), costimulatory enhancement, or NK cell recruitment (CD16).
Advantages of TsAbs: Dual TAA targeting improves tumor specificity and coverage while reducing antigen-loss escape; fusion of costimulatory domains such as CD28/4-1BB enhances T-cell proliferation and persistence; some also incorporate localized activation via cytokines (e.g., IL-15).
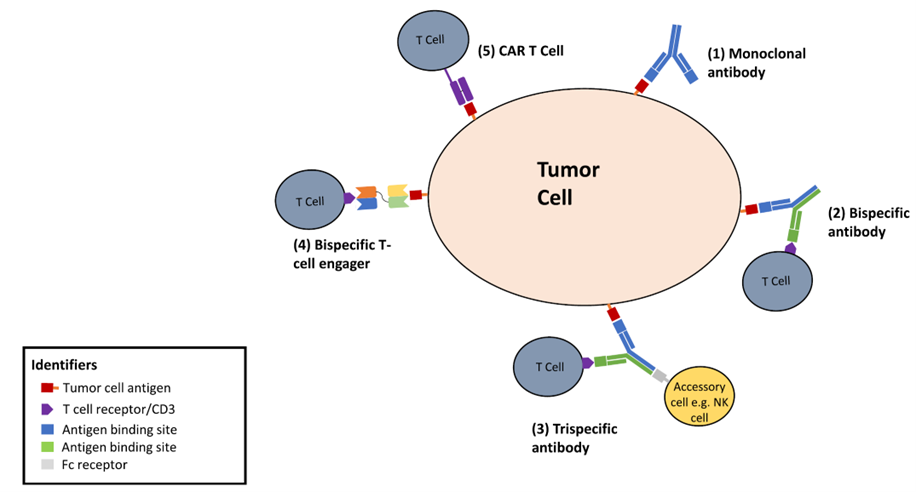
Figure 1. Binding modes of different forms of immunotherapy to tumor cell surface antigens
The concept of bispecific antibodies dates back to the 1960s, but true engineered production began in the 1980s with quadroma technology. Subsequently, platforms such as “knobs-into-holes,” CrossMab, and BiTE overcame challenges related to light/heavy chain mispairing, stability, and manufacturing. In 2014/2015, the first BsAb—blinatumomab (CD19 × CD3 BiTE)—was approved by the FDA and EMA for relapsed/refractory B-ALL, ushering in the clinical era. By the end of 2025, approximately 17 BsAbs had been approved worldwide, most of them for cancer treatment (primarily hematologic malignancies, with rapid expansion into solid tumors). New approvals in 2025, including linvoseltamab (BCMA × CD3 for RRMM), have further expanded treatment options.
Trispecific antibodies entered early-stage clinical development in the 2020s, aiming to address antigen escape and T-cell exhaustion associated with bispecific antibodies. Most are currently in Phase I/II trials, with some having received FDA Fast Track designation (e.g., IBI3003). Although none have yet received full approval, clinical data have already demonstrated potential to surpass bispecific antibodies.

Figure 2. Mechanisms of action of BsAbs in solid tumors and hematologic malignancies, along with various structural formats of BsAbs
JNJ-79635322/JNJ-5322 (BCMA × GPRC5D × CD3) in a Phase I trial (NCT05652335) demonstrated an overall response rate (ORR) of 86% in heavily pretreated RRMM patients (median 4 prior lines), reaching 100% at the recommended Phase 2 dose (RP2D), with 89% achieving ≥VGPR. In patients naïve to both BCMA and GPRC5D therapies, the ORR was 100%. Cytokine release syndrome (CRS) was mostly Grade 1 (no ≥Grade 3 events), supporting once-every-4-weeks (Q4W) subcutaneous administration with a safety profile superior to some bispecific antibodies.
IBI3003 (GPRC5D × BCMA × CD3) has received FDA Fast Track designation for RRMM. Other candidates such as SIM0500 and PIT565 are also advancing, showing potential to overcome bispecific antibody resistance.
As the foundational product of the BiTE platform, it is primarily used for relapsed/refractory B-cell acute lymphoblastic leukemia (R/R B-ALL). Its mechanism involves low-affinity CD3 engagement to redirect T cells, forming an immune synapse that induces tumor cell apoptosis. This combination established the clinical foundation of the BiTE platform and represents a milestone in translating multispecific antibodies from concept to practice.

Figure 3. Mechanism of action of blinatumomab
BCMA is a core target highly expressed on the surface of plasma cells in multiple myeloma (MM). These bispecific antibodies achieve efficient tumor killing through T-cell redirection. They have shown strong efficacy in triple-class exposed or later-line RRMM. This target class effectively addresses the heterogeneity of BCMA expression and serves as an important addition to standard treatment for RRMM.
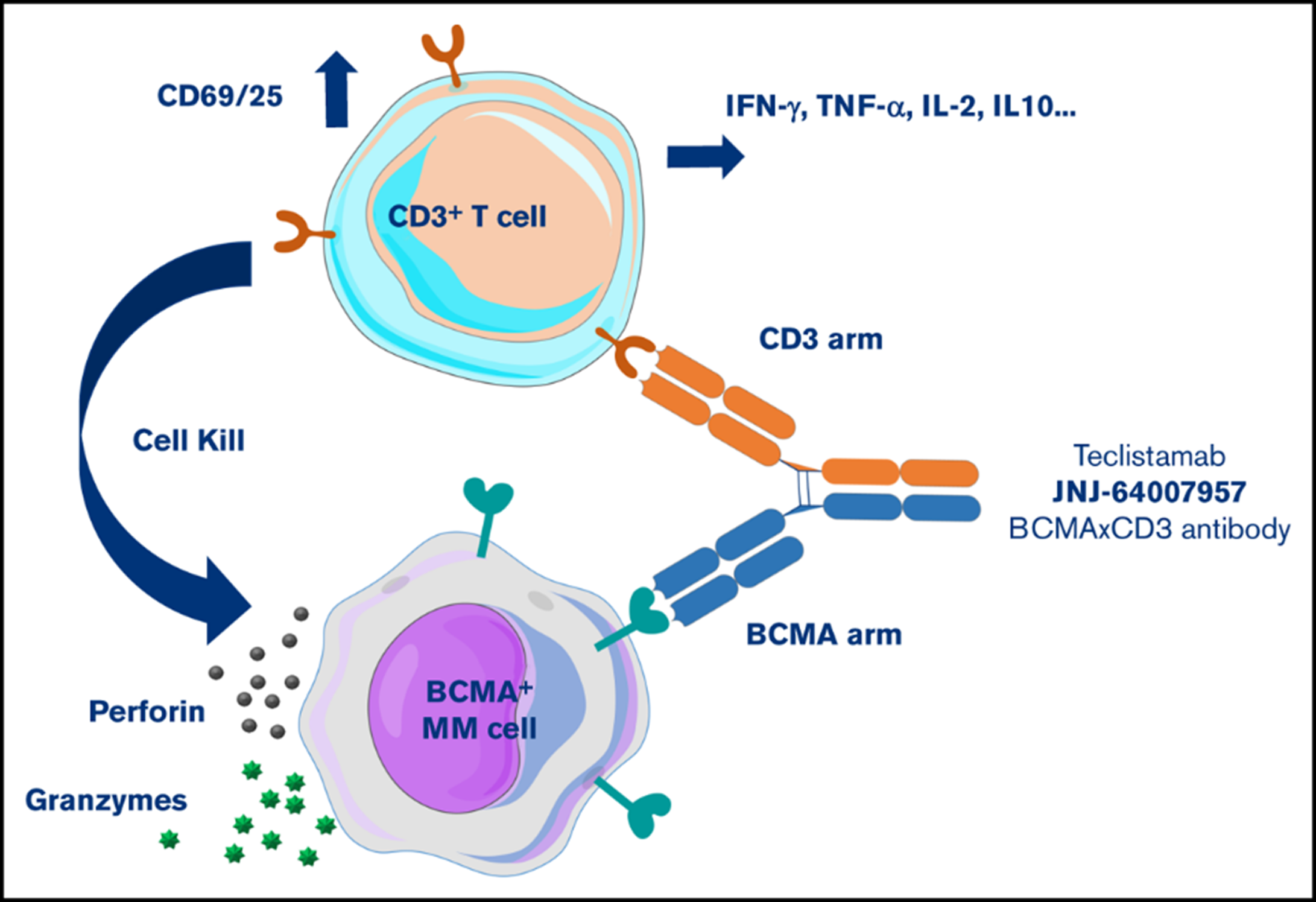
Figure 4. Mechanism of action of teclistamab
Designed for B-cell lymphomas (especially DLBCL and FL), these antibodies use a 2:1 asymmetric format (one CD20 arm and two CD3 arms) to enhance affinity for low-density CD20 antigens while retaining Fc-mediated effector functions. This combination effectively overcomes escape caused by downregulated CD20 expression through dual-target bridging and has become an important option for B-cell malignancies after second-line therapy.
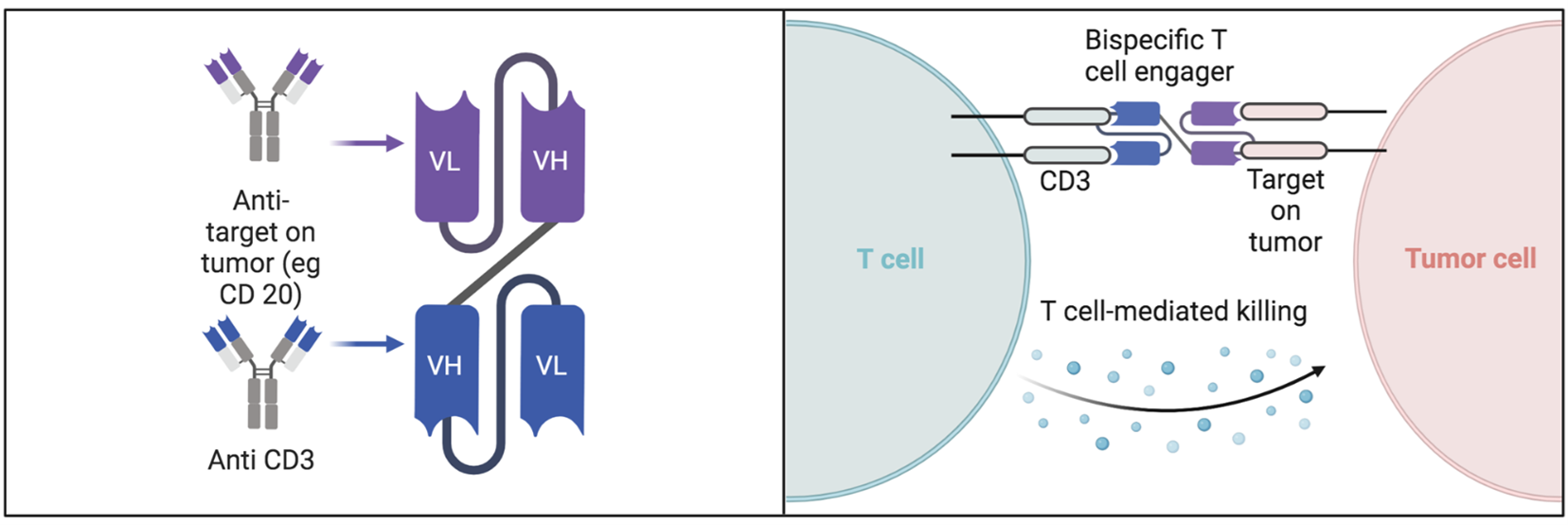
Figure 5. Mechanism of action of bispecific antibodies
GPRC5D is a novel surface marker in MM, particularly highly expressed in patients with BCMA resistance or low expression, effectively addressing antigen loss. The Monumental-1 trial confirmed that talquetamab achieved significant responses in triple-class exposed or later-line RRMM, especially in patients who failed prior BCMA-targeted therapies. This target is complementary to BCMA, forming a “dual insurance” strategy that further expands the treatment landscape for MM.
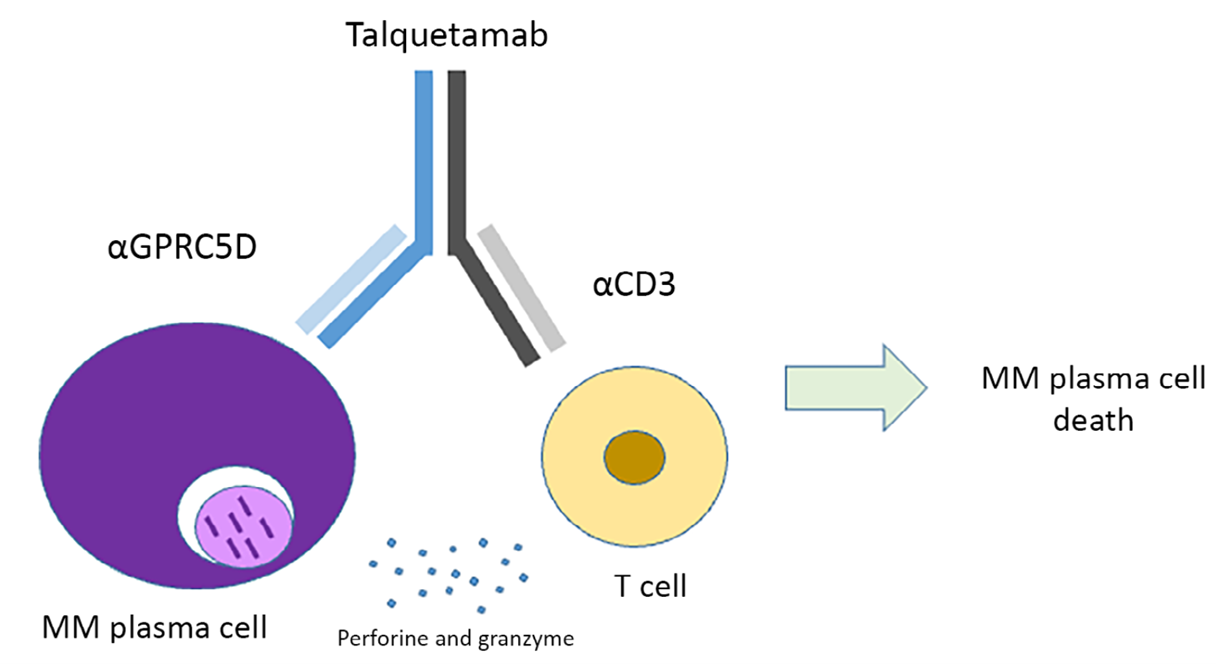
Figure 6. Talquetamab mediates T-cell activation and killing of multiple myeloma cells by simultaneously binding T-cell CD3 and plasma cell GPRC5D
The dual TAA + CD3 design simultaneously targets BCMA and GPRC5D, significantly improving tumor coverage and reducing the risk of antigen escape, while using a low-affinity CD3 domain to optimize safety. In a Phase I trial involving RRMM patients (median 4 prior lines), the RP2D (100 mg Q4W) achieved an ORR of 86% (75% ≥VGPR). In the subgroup naïve to both BCMA and GPRC5D therapies, the ORR reached 100% (89% ≥VGPR), with all patients maintaining response (median follow-up 8.5 months). CRS was predominantly Grade 1 (no ≥Grade 3 events), supporting monthly subcutaneous dosing with a safety profile superior to most bispecific antibodies. This design represents the next-generation paradigm of multispecific antibodies, with the potential to deliver CAR-T-like efficacy in an “off-the-shelf” format suitable for outpatient treatment.
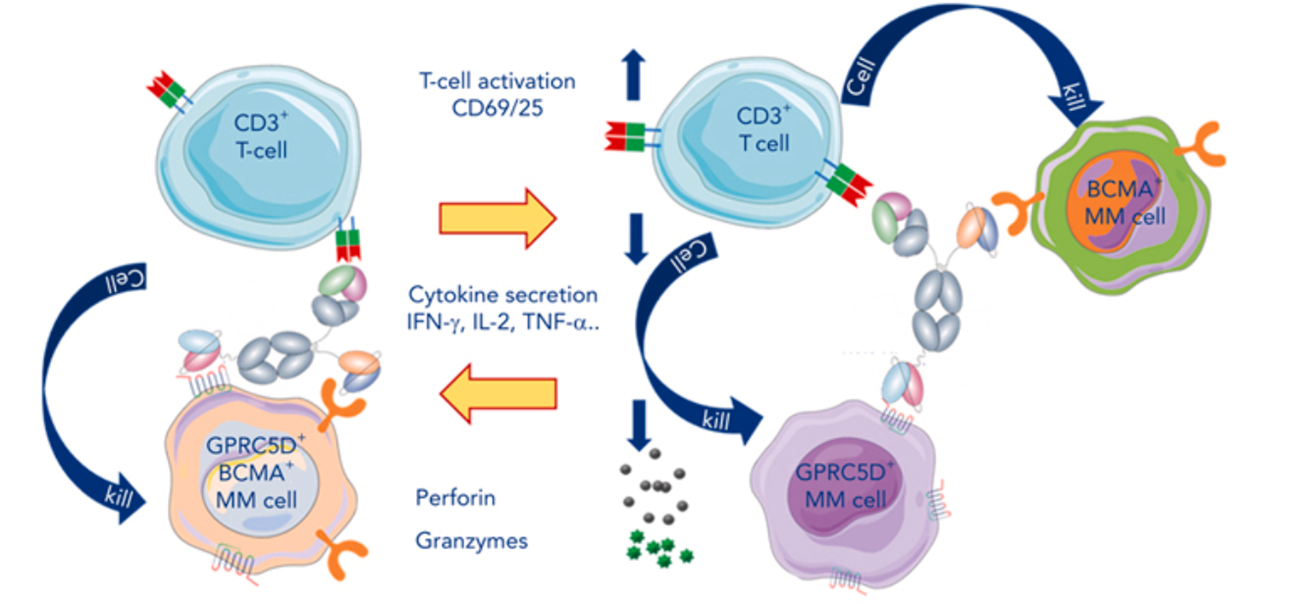
Figure 7. Recruitment of T cells to tumor cells via a trispecific antibody
During the clinical translation of bispecific and trispecific antibodies, safety and drug resistance remain key areas requiring optimization. Common immune-related adverse reactions include cytokine release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS), and cytopenias due to bone marrow suppression.
These toxicities have been significantly mitigated through advanced engineering strategies such as step-up dosing, Fc-silencing designs, protease-activatable masking peptides, and low-affinity CD3 binding domains, allowing most patients to tolerate treatment well.
The primary drivers of resistance are the loss of target antigens on tumor cells and exhaustion of effector T cells. Future strategies to address these challenges include combinations of BsAbs/TsAbs with CELMoD (cereblon E3 ligase modulators) agents, synergy with PD-1 immune checkpoint inhibitors, or further development of quad-specific antibody platforms to achieve multidimensional immune activation and tumor microenvironment remodeling.
Trispecific antibodies and next-generation multispecific antibodies are rapidly expanding from hematologic malignancies into solid tumors. Fixed-duration treatment, convenient subcutaneous injection regimens, and combination therapies with antibody-drug conjugates (ADCs) and personalized tumor vaccines are expected to become mainstream directions. In the future, more TsAbs are anticipated to receive full regulatory approval from agencies such as the FDA, fully ushering in the “multi-target synergy” era of precision cancer immunotherapy and offering cancer patients more durable and effective treatment options.
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

