AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2026-01-08 View volume: 334
Service Overview — AtaGenix provides end-to-end bispecific antibody (BsAb) custom development, supporting 15+ formats from design through purification and QC. Leveraging proprietary HEK293 and XtenCHO™ expression platforms with >90% project success rate, the service addresses key engineering challenges including chain mispairing, yield optimization, and format-specific purification — enabling reliable downstream functional validation and in vivo studies.
Bispecific antibodies simultaneously recognize two distinct targets, enabling mechanisms such as immune cell redirection and coordinated signal modulation that conventional monoclonal antibodies cannot achieve. The emphasis has shifted from whether BsAbs work to how they can be developed in a stable, safe, and reproducible manner — making a robust antibody engineering platform essential for translating bispecific concepts into reliable R&D outcomes.
A 2023 Lancet review (DOI: 10.1016/S0140-6736(23)00521-4) comprehensively summarizes the clinical progress of T cell–redirecting BsAbs in oncology. Several products have achieved FDA/EMA approval in hematological malignancies, with solid tumor exploration expanding. The review repeatedly emphasizes that antibody format and engineering design profoundly impact safety, half-life, dosing, and adverse event management — confirming that BsAb development is a highly integrated engineering process.
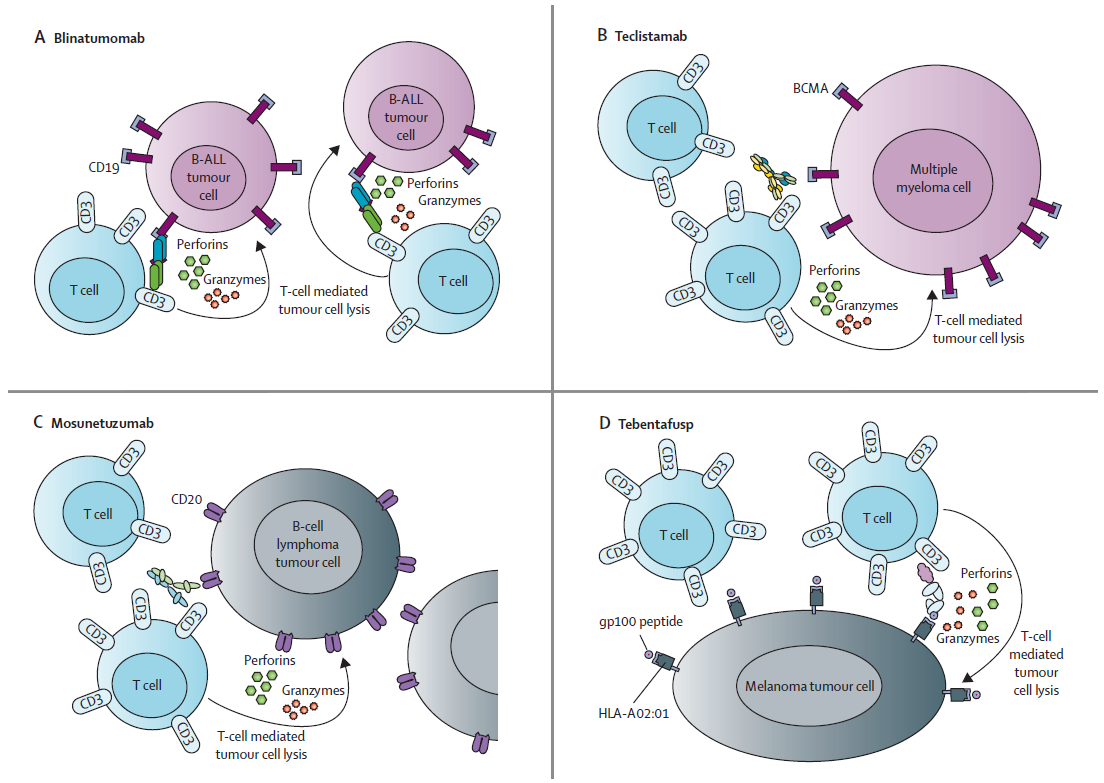
Fig. 1. Structures of approved T cell–redirecting bispecific antibody formats. Adapted from The Lancet 2023.
Mispairing Risks: Multi-chain/multi-arm architectures create heavy-light chain mispairing that must be engineered out.
Yield & Purity: Differential expression of antibody arms creates heterogeneous product pools requiring specialized purification.
Stability & Reproducibility: Structural complexity impacts protein stability and batch-to-batch consistency.
Format-Specific Requirements: Each BsAb architecture demands tailored expression systems and purification workflows.
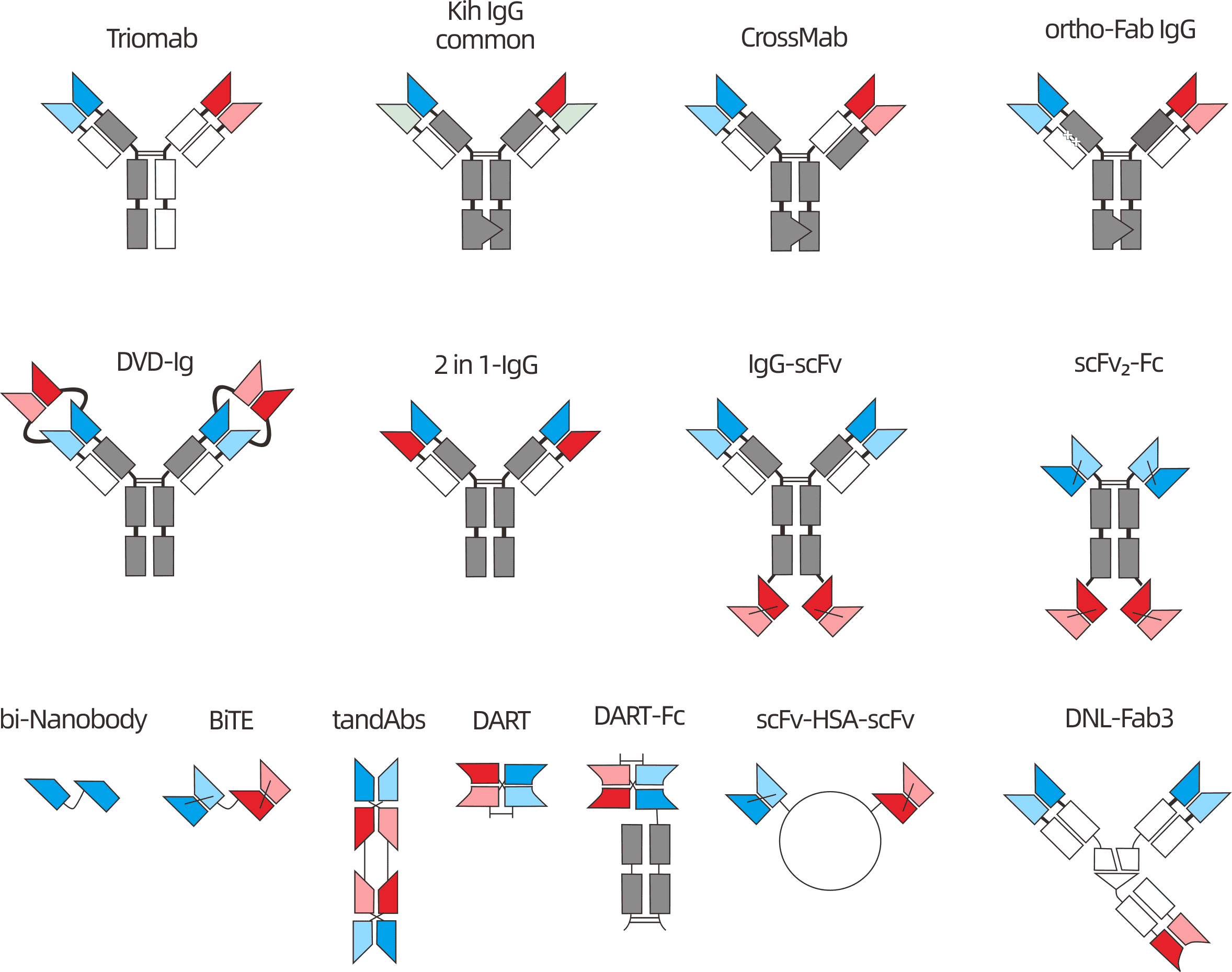
15+ BsAb Formats
BiTE, IgG-like, Fab-based, nanobody-based, multispecific
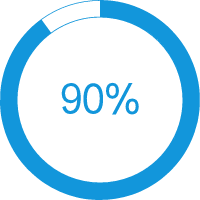
>90% Success Rate
Based on pre-agreed acceptance criteria

HEK293 / XtenCHO™
Proprietary optimized expression platforms

Multi-Strategy Purification
Tailored for complex antibody formats

Comprehensive QC
SDS-PAGE, SEC-HPLC, ELISA, endotoxin

Optimized Vectors
High-efficiency expression vector library
Antibody design strategies can be flexibly selected based on target combinations and research objectives. Currently supported formats include:
| Service Item | Client Provides | Service Content | Timeline | Deliverables |
|---|---|---|---|---|
| Bispecific Antibody Development | Antibody sequences | Gene synthesis & codon optimization, vector construction, expression & purification, QC analysis | 5–6 weeks | Purified antibody + technical service report |
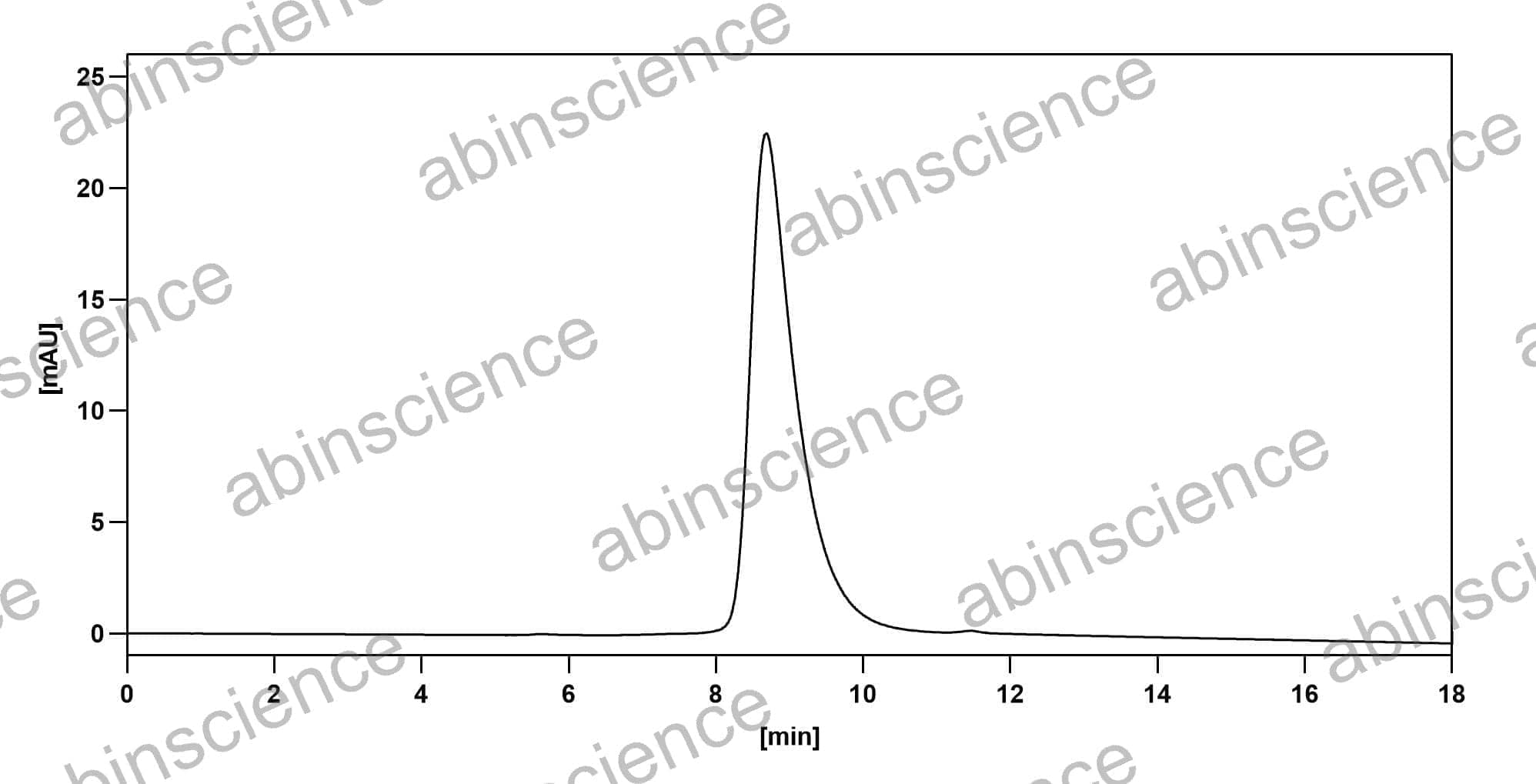

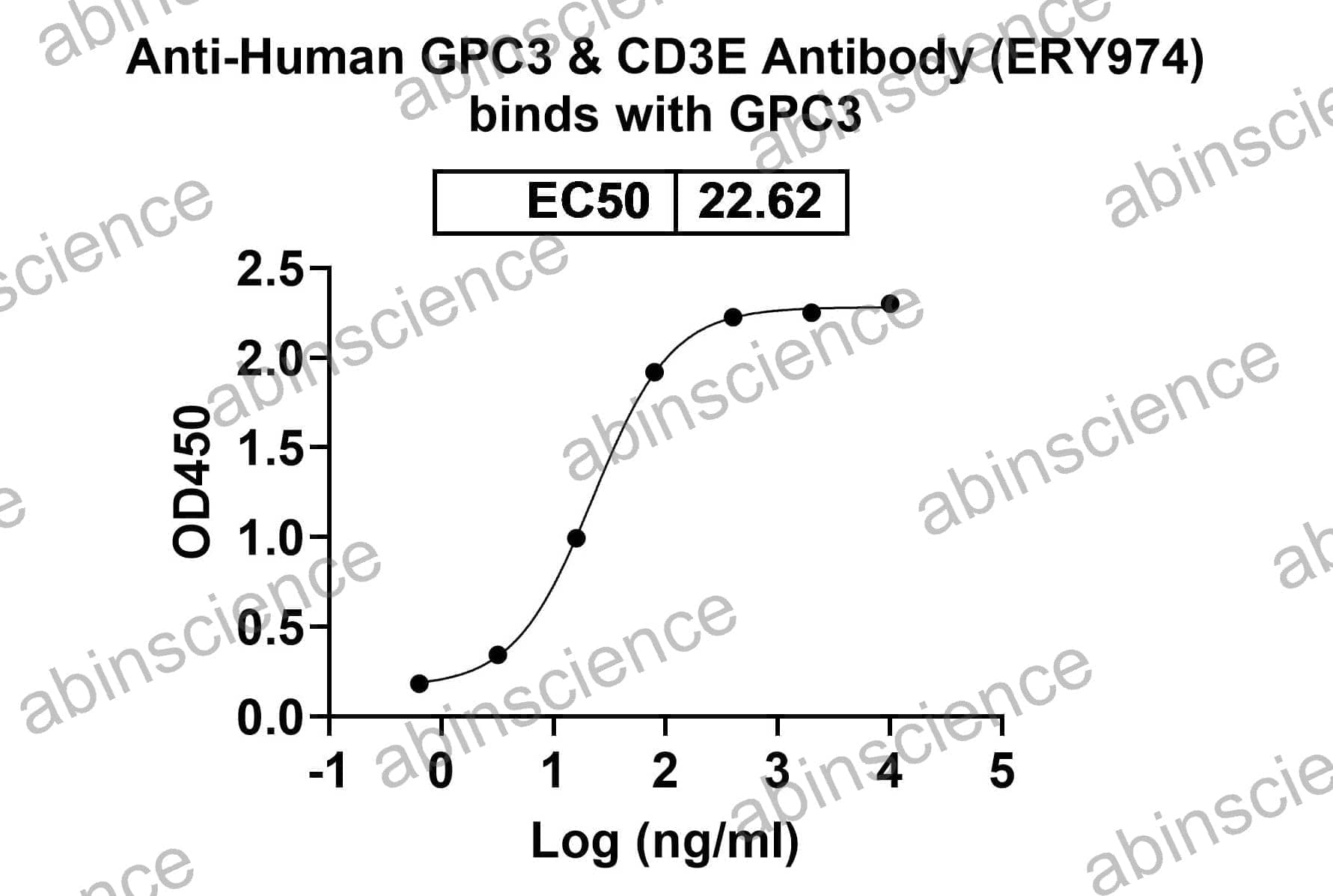
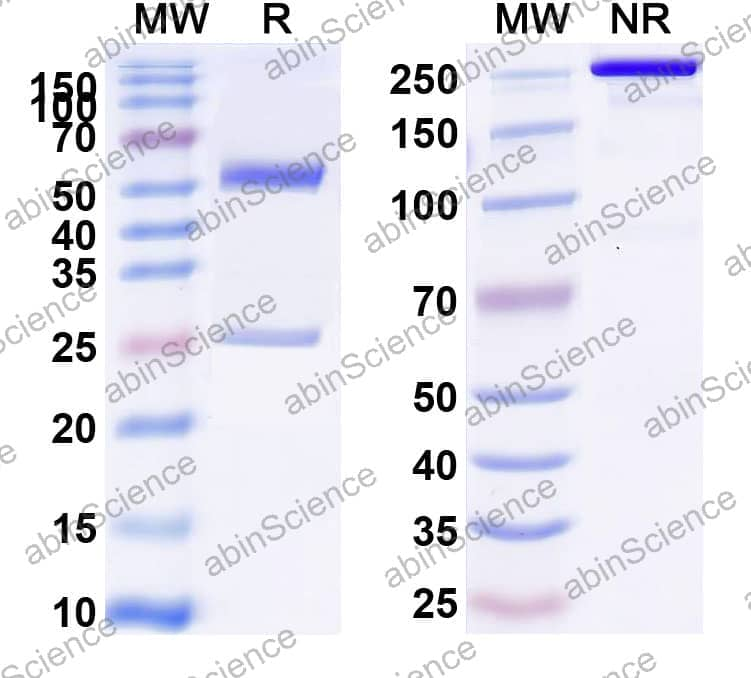
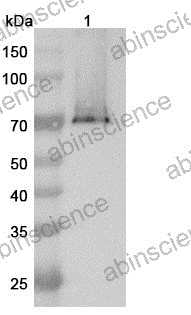
Ready to start your bispecific antibody project? Share your sequences and target pair — we'll recommend the optimal format and propose a development plan within 24 hours.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

