AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-12-12 View volume: 322
Project Snapshot — A Nature Microbiology study revealed that the E2 enzyme in E2-CBASS regulates cGAS via ubiquitination mimicry, promoting poly-cGAS formation and cGAMP production to confer anti-phage immunity in Serratia marcescens. AtaGenix provided a custom rabbit anti-cGAS polyclonal antibody that reliably distinguished cGAS monomers (46 kDa), cGAS-E2 complexes (64 kDa), and poly-cGAS (>100 kDa) in Western blot — enabling the study's core mechanistic validation.
Based on: Nature Microbiology, June 2024 — DOI: 10.1038/s41564-024-01684-z
Phage infection threatens bacterial survival, but the regulatory mechanisms of the prokaryotic cyclic-oligonucleotide-based anti-phage signalling system (CBASS) — especially the single E2 enzyme in E2-CBASS — remained unclear. This study focused on the E2-CBASS system from Serratia marcescens, using cryo-EM structural analysis and in vivo phage challenge experiments to define how E2 regulates cGAS activity.
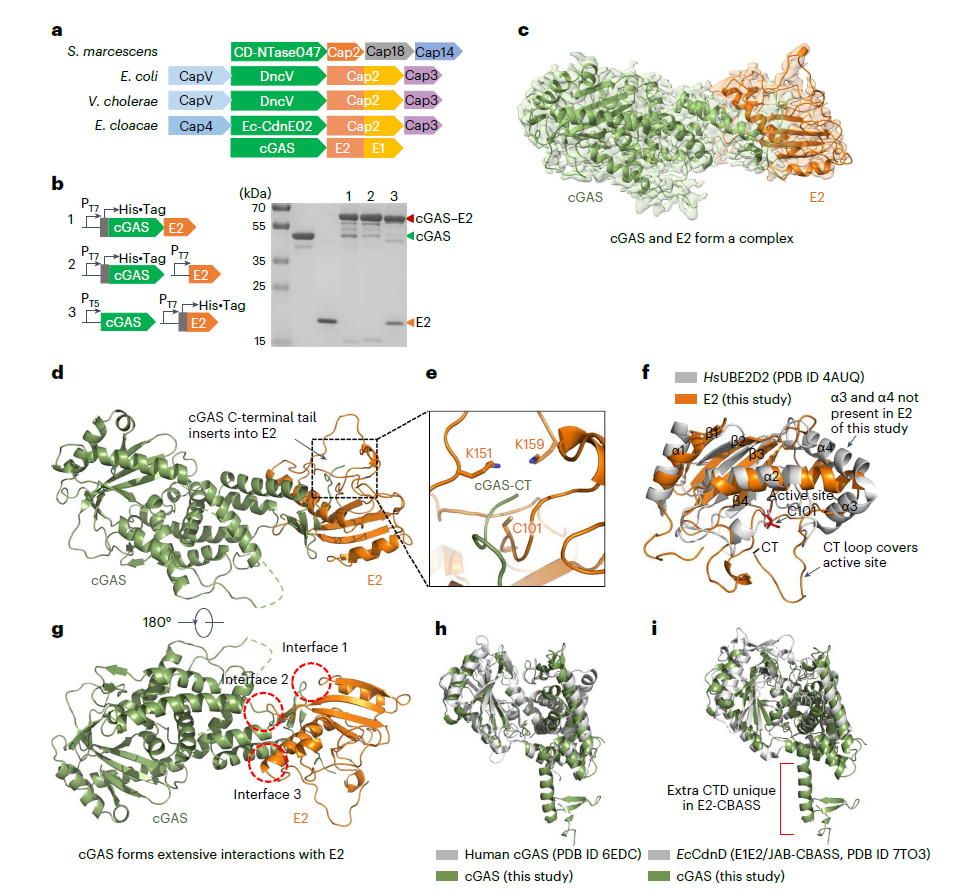
Fig. 1. Cryo-EM structure of the cGAS-E2 complex. E2 binds the cGAS C-terminal region via transient thioester (C101) and stable isopeptide (K159) bonds. Adapted from Nat Microbiol 2024.
Covalent Complex Formation: Cryo-EM confirmed E2 binds the cGAS C-terminal region. Critical residues (cGAS G406, E2 C101/K159) mediate two reversible covalent bonds: a transient thioester (cGAS Y405–E2 C101) and a stable isopeptide (cGAS Y405–E2 K159), regulated by pH.
Dual Enzymatic Activity: E2 exhibits both cysteine protease and ubiquitin ligase-like activity — cleaving the cGAS C-terminal G406-E407 peptide (>80% efficiency) and promoting poly-cGAS formation via inter-molecular isopeptide bonds.
Signal Amplification: Poly-cGAS is the active form, enhancing 3',2'-cGAMP synthesis by 2.3-fold. cGAMP activates the 2TM-SAVED effector protein, triggering membrane disruption and abortive infection to block phage replication.
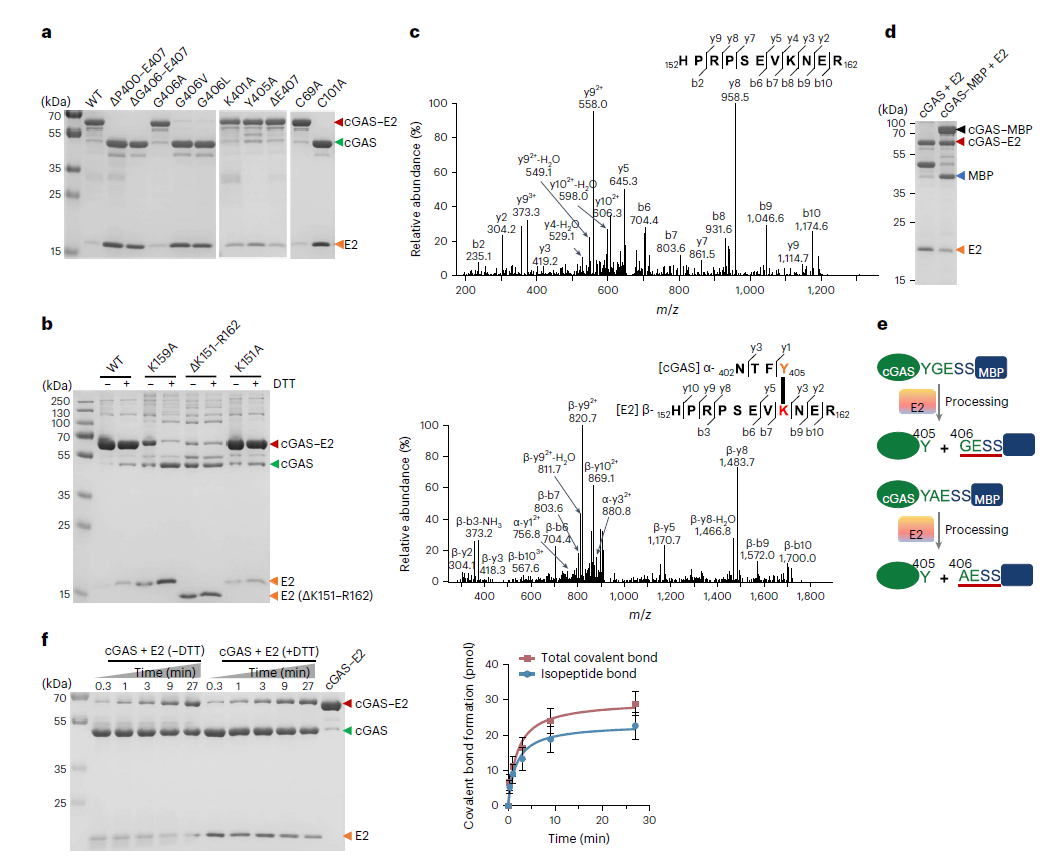
Fig. 2. Western blot detection of cGAS and its complexes using AtaGenix custom anti-cGAS pAb. Monomers (46 kDa), cGAS-E2 complexes (64 kDa), and poly-cGAS (>100 kDa) clearly resolved. Adapted from Nat Microbiol 2024.
In vivo experiments confirmed that E2-CBASS confers strong anti-phage immunity to E. coli against bacteriophage T4. Phage infection induces a drop in bacterial intracellular pH, activating E2 enzyme activity and driving poly-cGAS formation (4.2-fold increase 30 minutes post-infection, detected by the AtaGenix antibody). The generated cGAMP activates the 2TM-SAVED effector, triggering membrane disruption and abortive infection.
Nat Microbiol
Journal Published
2.3×
cGAMP Synthesis Boost
4.2×
Poly-cGAS Increase (30 min)
Cryo-EM
Structural Validation
Why This Matters
This study identifies the E2 enzyme as a multifunctional regulator (protease/E2/E3) in E2-CBASS, filling key gaps in understanding how prokaryotic anti-phage immunity operates at a molecular level. The discovery of ubiquitination mimicry in bacteria reveals unexpected evolutionary links between prokaryotic and eukaryotic innate immunity. The AtaGenix custom anti-cGAS polyclonal antibody was the essential detection tool — no commercial antibody could resolve cGAS monomers, complexes, and poly-cGAS simultaneously in Western blot, making this a de novo custom development critical to the study's conclusions.
Results may vary depending on target, antigen design, and project scope. All proprietary client information is subject to NDA.
Need custom antibodies against novel prokaryotic or viral targets for structural biology or mechanistic validation? AtaGenix delivers antigen-to-antibody programs with application-specific QC.
Talk to Technical SupportResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

