AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-08-24 View volume: 609
Project Snapshot — A 2022 Journal of Immunology study identified shrimp MANF (Mesencephalic Astrocyte-derived Neurotrophic Factor) as a conserved anti-inflammatory plasma protein that suppresses ERK/NF-κB signaling via receptor-type tyrosine phosphatase (RPTP) binding. AtaGenix provided phage display-derived monoclonal antibodies and CHO-expressed recombinant MANF that were central to the study's sandwich ELISA quantification and mechanistic validation.
Based on: Luo K, Chen Y, Wang F, J. Immunology, 2022 (DOI: 10.4049/jimmunol.2100595)
Inflammation is a double-edged sword: too little leaves the host vulnerable to infection; too much causes self-damage. In mammals, this balance is modulated by a complex network of cytokines, receptors, and intracellular signaling cascades. But which of these regulatory mechanisms are truly ancient and conserved — and which are evolutionary add-ons? Invertebrates like shrimp offer a stripped-down model system where the core immune logic is easier to dissect.
This study, using the Pacific white shrimp (Litopenaeus vannamei), discovered that MANF — previously known only as a vertebrate neurotrophic factor — functions as an LPS-induced anti-inflammatory plasma protein in invertebrates. MANF binds RPTP on hemocytes (the shrimp equivalent of macrophages), dephosphorylates ERK, and suppresses NF-κB (Dorsal) activation. The finding that this mechanism also operates when shrimp RPTP is expressed in human 293T cells suggests deep evolutionary conservation — with potential implications for neuroprotection and inflammation research in mammals.

Figure 1. MANF is conserved from invertebrates (shrimp) to vertebrates (human). Phylogenetic analysis and sequence alignment demonstrate high structural conservation, suggesting that MANF's anti-inflammatory function predates the vertebrate immune system.
Proving that MANF is a secreted anti-inflammatory factor — not just an intracellular stress-response protein — required quantifying its levels in shrimp plasma before and after LPS challenge. No commercial ELISA kits exist for shrimp MANF. The research team needed two critical custom reagents: (1) highly specific monoclonal antibodies against shrimp MANF for sandwich ELISA construction, and (2) recombinant MANF protein for functional studies (dose-response experiments on hemocyte ERK/NF-κB signaling).
Both reagents presented unique difficulties. Shrimp MANF had not been previously characterized as a protein, so no validated immunogens or expression constructs existed. The antibodies had to be specific enough to detect MANF in the complex background of shrimp hemolymph plasma, and the recombinant protein had to retain biological activity for cell-based signaling assays.
Using the AtaGenix-developed sandwich ELISA, the team showed that MANF levels in shrimp plasma increased significantly after LPS injection — the first evidence that MANF functions as a secreted immune mediator in any invertebrate species.
Recombinant MANF treatment of hemocytes reduced ERK phosphorylation and NF-κB (Dorsal) expression in a dose-dependent manner. RNA interference knockdown of RPTP abolished this effect, proving that MANF acts through RPTP-mediated dephosphorylation of ERK — a previously unknown signaling axis.
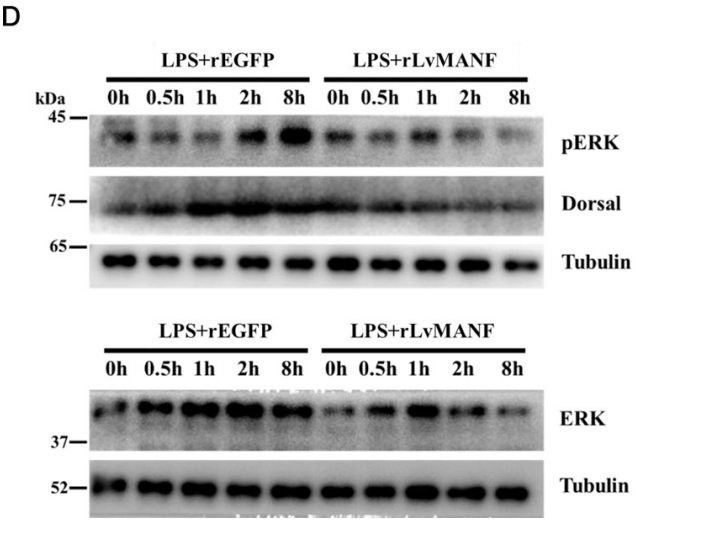
Figure 2. AtaGenix-produced recombinant MANF suppresses ERK phosphorylation and NF-κB (Dorsal) expression in shrimp hemocytes. RPTP knockdown abolishes the anti-inflammatory effect, confirming the MANF–RPTP–ERK signaling axis.
When shrimp RPTP was overexpressed in human 293T cells, MANF's effect on ERK switched from activation (via endogenous mammalian receptors) to inhibition (via RPTP) — demonstrating that the MANF–RPTP anti-inflammatory axis is functionally conserved across 600 million years of evolution.
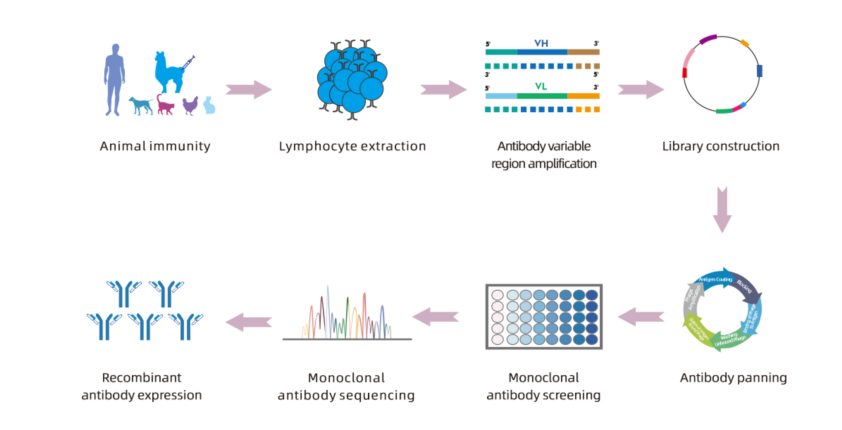
Figure 3. AtaGenix's workflow: phage display screening identified matched monoclonal antibody pairs, which were used to construct a sandwich ELISA for quantifying MANF levels in shrimp plasma. This assay provided the critical in vivo evidence that MANF is secreted as an anti-inflammatory factor after immune challenge.
Luo K, Chen Y, Wang F. Shrimp Plasma MANF Works as an Invertebrate Anti-Inflammatory Factor via a Conserved Receptor Tyrosine Phosphatase. The Journal of Immunology. 2022. DOI: 10.4049/jimmunol.2100595
Why This Matters
Studying novel immune mechanisms in non-model organisms requires building every reagent from scratch — no catalog antibodies, no commercial ELISA kits, no established expression constructs. This project is a textbook example of AtaGenix's integrated capability: phage display antibody discovery (because the target was completely novel), CHO expression for bioactive recombinant protein (because MANF's disulfide bonds demanded eukaryotic folding), and custom ELISA development (because no assay existed). The result was a Journal of Immunology paper that uncovered a 600-million-year-old anti-inflammatory mechanism — built entirely on custom reagents that didn't exist before AtaGenix made them.
This case study is based on a published research collaboration. Results may vary depending on target protein, antibody specificity requirements, and assay format. All proprietary client information is subject to NDA.
Studying a novel target with no existing reagents? AtaGenix builds the complete toolkit — monoclonal antibodies via phage display, bioactive recombinant protein, and custom immunoassays — so your biology doesn't wait for catalog availability.
Discuss Your Novel Target ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

