AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-07-21 View volume: 523
Project Snapshot — A research team studying METRNL's protective role in pancreatic β-cells needed high-purity, endotoxin-free recombinant protein validated for both in vivo diabetic mouse models and in vitro functional assays. AtaGenix delivered a fully characterized rMet protein via HEK293 expression, enabling a high-impact publication in Diabetologia (2025).
Based on: Diabetologia, 2025 — DOI: 10.1007/s00125-025-06459-7
Meteorin-like (METRNL) is an emerging secreted protein with a crucial role in safeguarding pancreatic β-cells, enhancing insulin secretion, and counteracting diabetes-associated metabolic stress. Recent studies have demonstrated that METRNL preserves β-cell identity by inhibiting β-to-α trans-differentiation, thereby slowing the progression of insulin resistance and diabetes. A landmark paper published in Diabetologia (2025, DOI: 10.1007/s00125-025-06459-7) highlighted its therapeutic potential in diabetes intervention.
The collaborating research team sought to investigate METRNL's protective effects under metabolic stress. To achieve this, they required recombinant METRNL (rMet) protein of exceptionally high quality: purity above 95%, endotoxin-free, and validated for both in vivo mouse models (including high-fat diet and db/db mice) and in vitro assays such as insulin secretion, Western blotting, IHC, and ELISA. In short, the team needed a reliable, ready-to-use protein that could seamlessly support both mechanistic studies and translational research.
Folding & Bioactivity: METRNL's tendency to misfold during recombinant expression often reduces bioactivity.
In Vivo Safety: Strict endotoxin-free requirements had to be met for in vivo diabetic mouse models.
Stability: Ensuring protein stability in metabolically stressed diabetic models was critical.
Consistent Supply: High-yield, high-purity expression systems needed to be optimized for reproducible production.
Smart Design & Expression: A HEK293 mammalian cell expression system was chosen to ensure proper folding and post-translational modifications. Codon optimization and a His-tag strategy enhanced both yield and purification efficiency.
Robust Purification: Multi-step purification combining Ni-NTA affinity and ion-exchange chromatography delivered protein purity above 95%. Endotoxin testing (LAL assay) confirmed levels below detection limits, meeting in vivo safety standards.
Comprehensive Validation: Identity was confirmed with METRNL-specific antibodies via Western blot, purity by SDS-PAGE, and functional concentration/activity by ELISA. Functional assays in INS-1 β-cells and mouse islets demonstrated enhanced insulin secretion and regulation of key β-cell genes (Ins1, Ins2, Pdx1).
End-to-End Support: Beyond delivery, AtaGenix provided QC reports, application protocols, and technical guidance to ensure smooth integration into the team's experimental pipeline.
With AtaGenix's rMet protein, the research team successfully demonstrated improved glucose clearance and plasma insulin levels in both high-fat diet and db/db mouse models. Importantly, rMet inhibited β-to-α trans-differentiation, reinforcing β-cell identity under metabolic stress. These findings confirmed the role of METRNL–c-Jun signaling in diabetes pathophysiology and opened new avenues for developing METRNL as a therapeutic target.
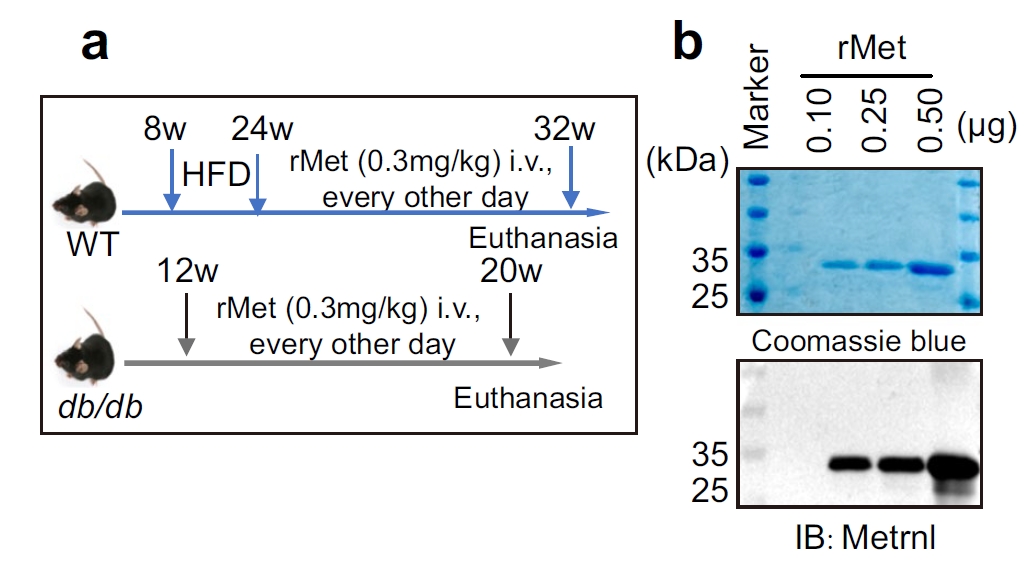
Fig. 1. In vivo validation of rMet protein in diabetic mouse models. Administration of AtaGenix-produced recombinant METRNL improved glucose clearance and plasma insulin levels. Adapted from Diabetologia 2025.
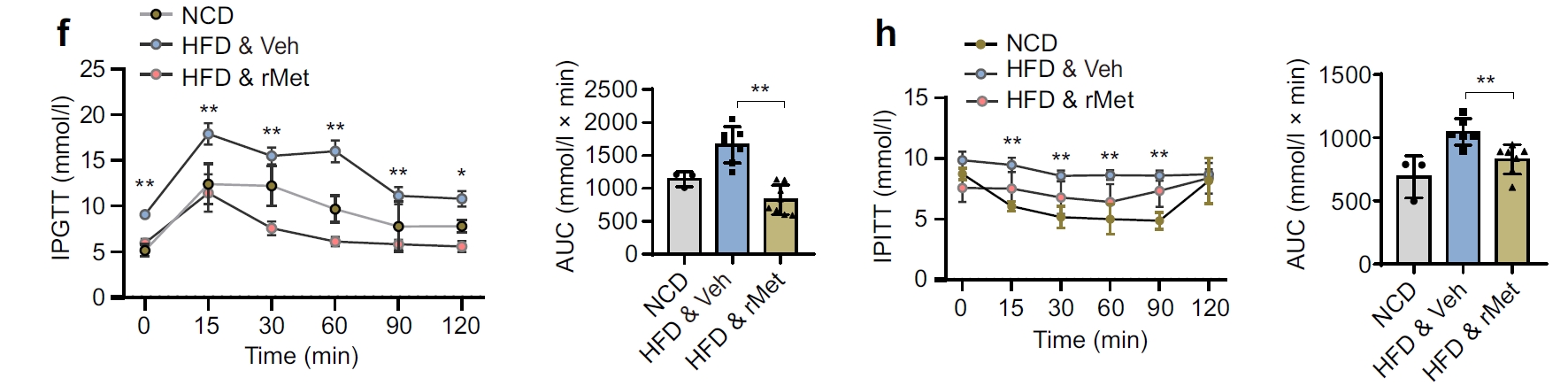
Fig. 2. METRNL functional assays in INS-1 β-cells and mouse islets demonstrated enhanced insulin secretion and upregulation of key β-cell identity genes (Ins1, Ins2, Pdx1). Adapted from Diabetologia 2025.
>95%
Protein Purity
Endotoxin (LAL)
In Vivo
Validated (HFD + db/db)
Diabetologia
Journal Published
Why This Matters
This study establishes METRNL as a protective factor for pancreatic β-cells, demonstrating that recombinant METRNL can inhibit β-to-α trans-differentiation and improve insulin secretion under metabolic stress. The AtaGenix-produced rMet protein was the core reagent enabling both in vivo validation (HFD and db/db mouse models) and in vitro mechanistic assays — requiring mammalian expression for proper folding, endotoxin-free purity for in vivo safety, and functional validation across multiple assay platforms.
Diabetologia, 2025. DOI: 10.1007/s00125-025-06459-7
For research use only. Results may vary depending on target protein, model system, and project scope. All proprietary client information is subject to NDA.
Need reliable recombinant proteins for diabetes or metabolic disease research? AtaGenix delivers customized, high-quality proteins with full validation support — from HEK293/CHO expression through in vivo-grade QC.
Discuss Your Protein ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

