AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-06-20 View volume: 457
Project Snapshot — Researchers at the Chinese Academy of Medical Sciences (Kunming) needed high-purity HSV-1 glycoprotein B (gB) and its prefusion mutant gB H516P for a subunit vaccine immunogenicity study. AtaGenix delivered both proteins via CHO mammalian expression with nickel column purification, enabling ELISpot and ELISA validation that led to a publication in Vaccine (2025).
Based on: Hu J et al., Vaccine, 2025 — DOI: 10.1016/j.vaccine.2025.127241
Herpes simplex virus 1 (HSV-1) infects over 3.7 billion people globally, causing mucocutaneous lesions and, in rare cases, life-threatening encephalitis. Despite decades of research, no approved prophylactic vaccine exists. A key obstacle has been generating antigens that reliably induce the cellular immune responses needed to control latent HSV-1 infections.
A 2025 study published in Vaccine (DOI: 10.1016/j.vaccine.2025.127241) by Jingping Hu and colleagues investigated whether stabilizing HSV-1 fusion glycoprotein B in its prefusion conformation (H516P mutation) would improve immunogenicity in mRNA and subunit vaccine formats. The study found no significant difference between wild-type gB and gB H516P, but demonstrated that subunit vaccines adjuvanted with QS-21 and CpG ODNs induced robust T-cell responses — a promising finding for HSV-1 vaccine development.
Dual Variant Production: Two closely related gB variants (wild-type and H516P mutant) needed to be produced under identical conditions for a clean head-to-head immunological comparison.
Glycosylation Consistency: Batch-to-batch glycosylation variability had to be minimized so that differences in immune response would not be confounded by production artifacts.
Structural Constraint: The H516P prefusion lock introduced a structural constraint that risked affecting expression levels and protein stability.
Assay-Grade Purity: High purity was essential for accurate ELISpot and ELISA readouts, with proper conformational epitope preservation.
Expression System: Chinese hamster ovary (CHO) cells were selected over HEK293 for this project — CHO provides more consistent glycosylation profiles across batches, which was critical for head-to-head comparison of two closely related protein variants.
Construct Design: Both gB and gB H516P were expressed with C-terminal His-tags. Codon optimization for CHO maximized translational efficiency.
Purification: Nickel column (Ni-NTA) affinity chromatography as the primary capture step, followed by polishing to achieve high purity. Both variants were processed under identical conditions to eliminate batch-to-batch variability.
QC & Validation: SDS-PAGE confirmed molecular weight and purity. The research team subsequently validated both proteins in ELISpot (measuring IL-2 and IFN-γ production) and ELISA (measuring gB-specific IgG titers), confirming functional suitability as assay antigens.

Fig. 1. Study design and immune response overview. The research compared mRNA and subunit vaccine formats using wild-type gB and prefusion-stabilized gB H516P with QS-21/CpG ODN adjuvants. Adapted from Vaccine 2025.
Both AtaGenix-produced proteins performed equivalently in immunological assays, which was itself a key finding — it demonstrated that the H516P prefusion lock did not improve immunogenicity over wild-type gB in these vaccine formats. More importantly for the vaccine field, the subunit formulations adjuvanted with QS-21 and CpG ODNs induced strong IFN-γ and IL-2 T-cell responses, suggesting that cellular immunity (critical for controlling latent herpesvirus) can be effectively elicited with properly formulated protein-based vaccines.
For the client, having access to well-matched, high-purity protein pairs allowed a clean head-to-head comparison that strengthened their publication and informed next-stage vaccine design decisions.
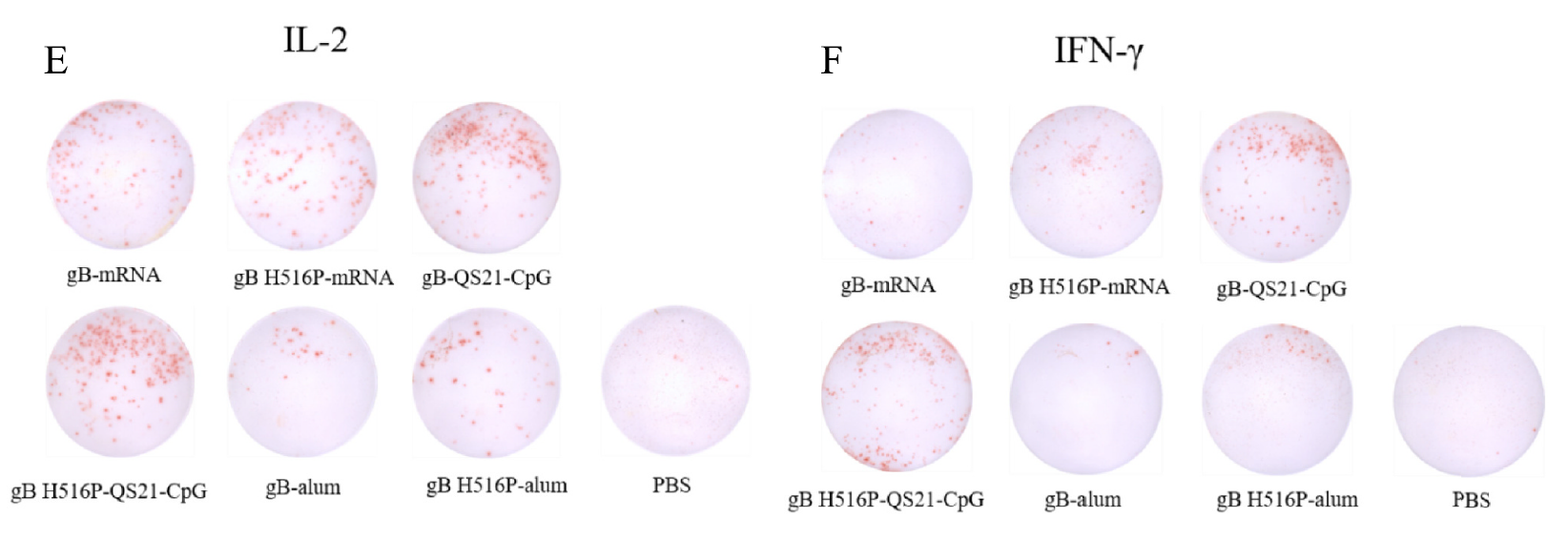
Fig. 2. ELISpot validation using AtaGenix-produced gB proteins. Both wild-type gB and gB H516P showed comparable T-cell stimulation, confirming functional equivalence as assay reagents. Adapted from Vaccine 2025.
CHO
Expression System
2
Matched Protein Variants
ELISpot
Functionally Validated
Vaccine
Journal Published
Why This Matters
Vaccine antigen studies that compare wild-type vs. engineered variants require production-matched protein pairs — any difference in purity, glycosylation, or folding can confound immunological readouts. This project demonstrates AtaGenix's ability to deliver parallel protein batches under identical conditions, giving researchers confidence that observed differences (or equivalences) in immune response are biologically meaningful, not artifacts of inconsistent protein quality.
For research use only. Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
Developing subunit vaccines or need matched antigen pairs for immunogenicity studies? AtaGenix provides end-to-end protein expression with batch-to-batch consistency for reliable comparative data.
Discuss Your Vaccine Protein ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

