AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-06-20 View volume: 492
Project Snapshot — Naval Medical University researchers needed matched sets of GST-tagged RMP variants and His-tagged IKKβ to map a previously unknown protein-protein interaction in sepsis-driven inflammation. AtaGenix delivered four high-purity E. coli-expressed proteins — including a critical S439A point mutant — that enabled SPR, GST-pulldown, and kinase assays, culminating in a publication in Cell Communication and Signaling (2025).
Based on: Pang S-J et al., Cell Communication and Signaling, 2025 — DOI: 10.1186/s12964-025-02278-w
Sepsis kills over 14 million people annually. At its core, sepsis involves a runaway inflammatory cascade driven by NF-κB signaling in macrophages. While the pathway from TLR4 activation to IκB kinase β (IKKβ) phosphorylation is well characterized, the endogenous "brakes" on this system remain poorly understood. Identifying these negative regulators could open new therapeutic avenues beyond conventional immunosuppression.
A 2025 study by Shu-jie Pang and colleagues at Naval Medical University, published in Cell Communication and Signaling (DOI: 10.1186/s12964-025-02278-w), identified RNA polymerase II subunit 5-mediating protein (RMP) as one such brake. RMP binds directly to IKKβ and recruits protein phosphatase 2A (PP2A), dephosphorylating IKKβ and dampening NF-κB-driven inflammation. Crucially, a single point mutation (S439A) at RMP's phosphorylation site enhanced its binding to IKKβ — a finding that required carefully matched recombinant proteins to demonstrate convincingly.

Fig. 1. Proposed mechanism: RMP binds IKKβ and recruits PP2A to dephosphorylate IKKβ, suppressing TLR4-induced NF-κB activation in macrophages during sepsis. The S439A mutation enhances IKKβ binding affinity. Adapted from Cell Commun Signal 2025.
Four Parallel Constructs: GST alone (negative control), GST-RMP wild-type, GST-RMP-S439A (phospho-dead mutant), and His-IKKβ — all had to be produced and purified under identical conditions.
Single-Residue Sensitivity: The S439A mutation produces subtle binding differences only detectable when production variables are eliminated.
Dual-Tag Strategy: GST tags for pulldown and His tag for orthogonal Ni-NTA purification and SPR chip immobilization required careful design.
Multi-Assay Compatibility: Proteins needed to perform in SPR, GST-pulldown, and kinase activity assays without buffer or purity artifacts.
Expression System: E. coli was the clear choice — RMP and IKKβ are intracellular signaling proteins that do not require glycosylation. The speed and yield of prokaryotic expression allowed all four constructs to be produced in a single batch cycle.
Site-Directed Mutagenesis: The S439A mutation was introduced by AtaGenix's molecular biology team with full sequence verification, ensuring the mutant differed from wild-type by exactly one codon.
Parallel Purification: All four proteins were purified under identical buffer and chromatography conditions (GST: glutathione agarose; His: Ni-NTA), eliminating purification-related variability from downstream comparisons.
QC: SDS-PAGE and Western Blot confirmed identity, purity, and intact tags for all constructs before shipment.
Direct binding confirmed by SPR: Real-time SPR (OpenSPR) demonstrated that RMP-WT binds IKKβ with measurable affinity, and the S439A mutant showed enhanced binding — providing the first biophysical evidence for this interaction.
GST-pulldown validated the interaction: GST-RMP-WT and GST-RMP-S439A both pulled down His-IKKβ, with the mutant showing stronger co-purification. GST alone showed no binding, confirming specificity.
Kinase inhibition quantified: ADP-Glo assays showed that RMP suppresses IKKβ kinase activity, consistent with its proposed role as a negative regulator of NF-κB signaling.
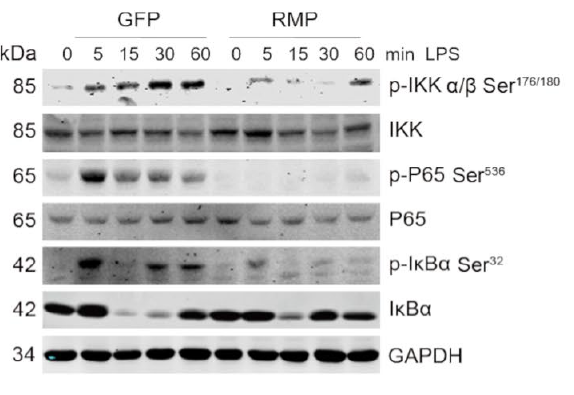
Fig. 2. SPR analysis of RMP–IKKβ binding. Real-time sensorgrams demonstrate direct interaction between His-IKKβ and GST-RMP proteins, with the S439A mutant showing enhanced binding. Adapted from Cell Commun Signal 2025.
Fig. 3. GST-pulldown assay. GST-RMP-WT and GST-RMP-S439A both co-purified with His-IKKβ, while GST alone did not. The S439A mutant showed stronger binding, consistent with SPR data. Adapted from Cell Commun Signal 2025.
4
Protein Constructs
E. coli
Expression System
SPR
Biophysically Validated
Cell Commun Signal
Journal Published
Why This Matters
Protein-protein interaction studies that compare wild-type vs. point mutants demand exceptional consistency between reagents. A single-residue change like S439A can produce subtle binding differences that are only detectable when production variables are eliminated. This project illustrates how AtaGenix's parallel expression pipeline — same host, same conditions, same purification — gives researchers the confidence to attribute observed differences to biology, not batch variation.
For research use only. Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
Need matched protein sets for interaction studies, kinase assays, or structural biology? AtaGenix delivers parallel-produced wild-type and mutant proteins under identical conditions.
Discuss Your Protein Interaction ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

