AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2025-06-20 View volume: 673
Project Snapshot — Researchers at Huazhong University of Science and Technology needed SPR-grade PRPF19 protein to test whether the natural compound berberine directly binds this E3 ligase — a critical experiment for establishing PRPF19 as a druggable target in diabetic kidney disease. AtaGenix delivered high-purity PRPF19 (1.67 mg/mL) that enabled Biacore T200 SPR confirmation of the berberine–PRPF19 interaction, contributing to a publication in Cell Communication and Signaling (2025).
Based on: He Q et al., Cell Communication and Signaling, 2025 — DOI: 10.1186/s12964-025-02253-5
Diabetic nephropathy (DN) is the leading cause of end-stage renal disease worldwide. While early-stage DN is driven by hemodynamic and metabolic factors, late-stage progression involves a less understood mechanism: ferroptosis — iron-dependent, lipid peroxidation-driven cell death in renal tubular epithelial cells (RTECs). The molecular switch controlling this process remained elusive until this study.
A 2025 study by Qiongyao He and colleagues, published in Cell Communication and Signaling (DOI: 10.1186/s12964-025-02253-5), identified PRPF19 as that switch. Under diabetic conditions, PRPF19 (an E3 ubiquitin ligase) tags the vitamin D receptor (VDR) for proteasomal degradation. Without VDR, the anti-ferroptosis enzyme GPX4 is downregulated, leaving RTECs vulnerable to iron-induced lipid peroxidation. The team then showed that berberine (BBR), a natural alkaloid, directly binds PRPF19 and inhibits its E3 ligase activity — stabilizing VDR and rescuing GPX4 expression. But proving "direct binding" required one critical experiment: SPR with purified PRPF19.

Fig. 1. Proposed mechanism: In diabetic conditions, PRPF19 ubiquitinates VDR for degradation, reducing GPX4 and triggering ferroptosis in RTECs. Berberine directly binds PRPF19 to block this cascade. Adapted from Cell Commun Signal 2025.
SPR-Grade Purity: Biacore T200 demands protein of exceptional quality — aggregates produce kinetic artifacts, and contaminants distort binding curves.
Accurate Concentration: Meaningful KD calculation requires precisely quantified protein for chip immobilization and analyte titration series.
Intact E3 Ligase Domain: Full-length PRPF19 had to retain its native fold for biologically relevant binding measurements.
Sufficient Quantity: Replicate injections across multiple berberine concentrations required ample protein supply beyond typical research-grade amounts.
Expression System: Optimized recombinant expression to produce full-length PRPF19 with intact E3 ligase domain architecture. The system was selected to maximize soluble yield while preserving the protein's native fold.
Purification for SPR: Multi-step chromatography was tailored to eliminate aggregates and co-purifying contaminants that would compromise Biacore sensorgram quality. Final protein was delivered at 1.67 mg/mL — well above the threshold for amine-coupling chip immobilization and multi-concentration analyte series.
QC Package: SDS-PAGE confirmed identity and purity. Concentration was verified by Bradford/BCA to ensure accurate SPR data fitting. The protein was additionally validated by the client's own Western Blot and immunoprecipitation assays, confirming functional integrity.
The AtaGenix-produced PRPF19 enabled the pivotal experiment in the study: Biacore T200 SPR confirmed that berberine directly binds PRPF19 in a concentration-dependent manner, with clear association and dissociation kinetics. This was the key piece of evidence elevating the finding from "berberine reduces PRPF19-mediated VDR degradation" (correlative) to "berberine physically engages PRPF19" (mechanistic).
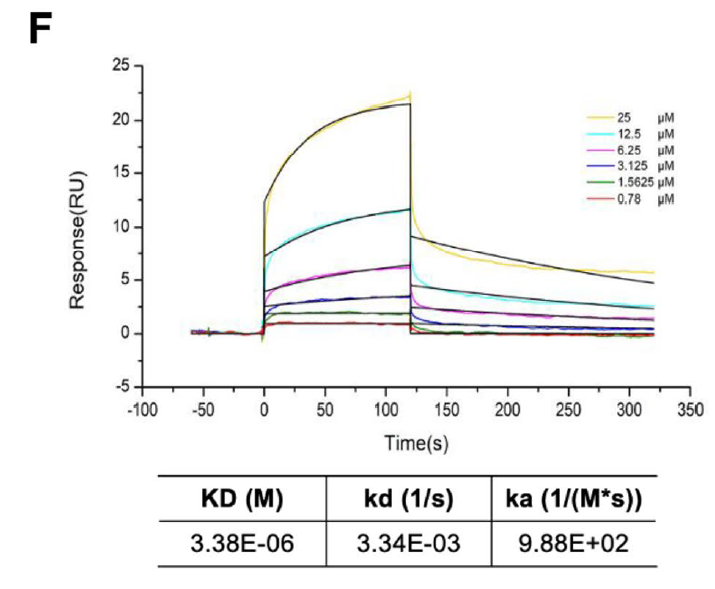
Fig. 2. Biacore T200 SPR analysis. Berberine binds to immobilized AtaGenix-produced PRPF19 with concentration-dependent kinetics, confirming direct physical interaction. Adapted from Cell Commun Signal 2025.
With direct binding established, the study went on to show that berberine treatment stabilized VDR, restored GPX4 expression, and reduced ferroptotic kidney damage in both cell models and diabetic mice — positioning the PRPF19–VDR axis as a promising therapeutic target for DN.
1.67
mg/mL Delivered
Biacore
T200 SPR Validated
WB + IP
Functionally Confirmed
Cell Commun Signal
Journal Published
Why This Matters
In drug target validation, the difference between "correlative" and "mechanistic" evidence often comes down to one experiment: a direct binding assay. For small-molecule-to-protein interactions, SPR is the gold standard — but it demands protein quality that many in-house labs cannot reliably produce. This project shows how outsourcing the critical reagent to AtaGenix allowed the research team to focus on biology while ensuring their binding data would withstand peer review scrutiny.
For research use only. Results may vary depending on target protein, construct design, and project scope. All proprietary client information is subject to NDA.
Need SPR-grade proteins for drug target validation or binding studies? AtaGenix delivers purified proteins optimized for Biacore, OpenSPR, and Octet platforms.
Discuss Your SPR Protein ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

