AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-11-04 View volume: 926
Project Snapshot — A client needed recombinant antibody expressed, purified, and functionally validated as a ready-to-use research reagent. AtaGenix delivered high-purity antibody with confirmed antigen specificity — demonstrating a stable, scalable workflow from gene to assay-grade material with comprehensive QC at every stage.
Recombinant antibody expression offers advantages that traditional hybridoma supernatant cannot match: defined sequence, batch-to-batch consistency, scalable production, and the ability to engineer format (IgG, Fab, scFv) and species (chimeric, humanized) as needed. But these advantages only materialize if the expression and purification workflow delivers antibody that is pure, correctly folded, and functionally active.
AtaGenix's recombinant antibody expression pipeline covers the full workflow: codon-optimized gene synthesis, vector construction (heavy + light chain co-expression), mammalian transient or stable expression, Protein A/G affinity purification, and a multi-assay QC panel that verifies not just purity but biological function. The goal is to deliver antibody that works in the client's assay on the first attempt — not just a protein that looks correct on a gel.
Every recombinant antibody produced by AtaGenix undergoes a standardized QC panel before release:
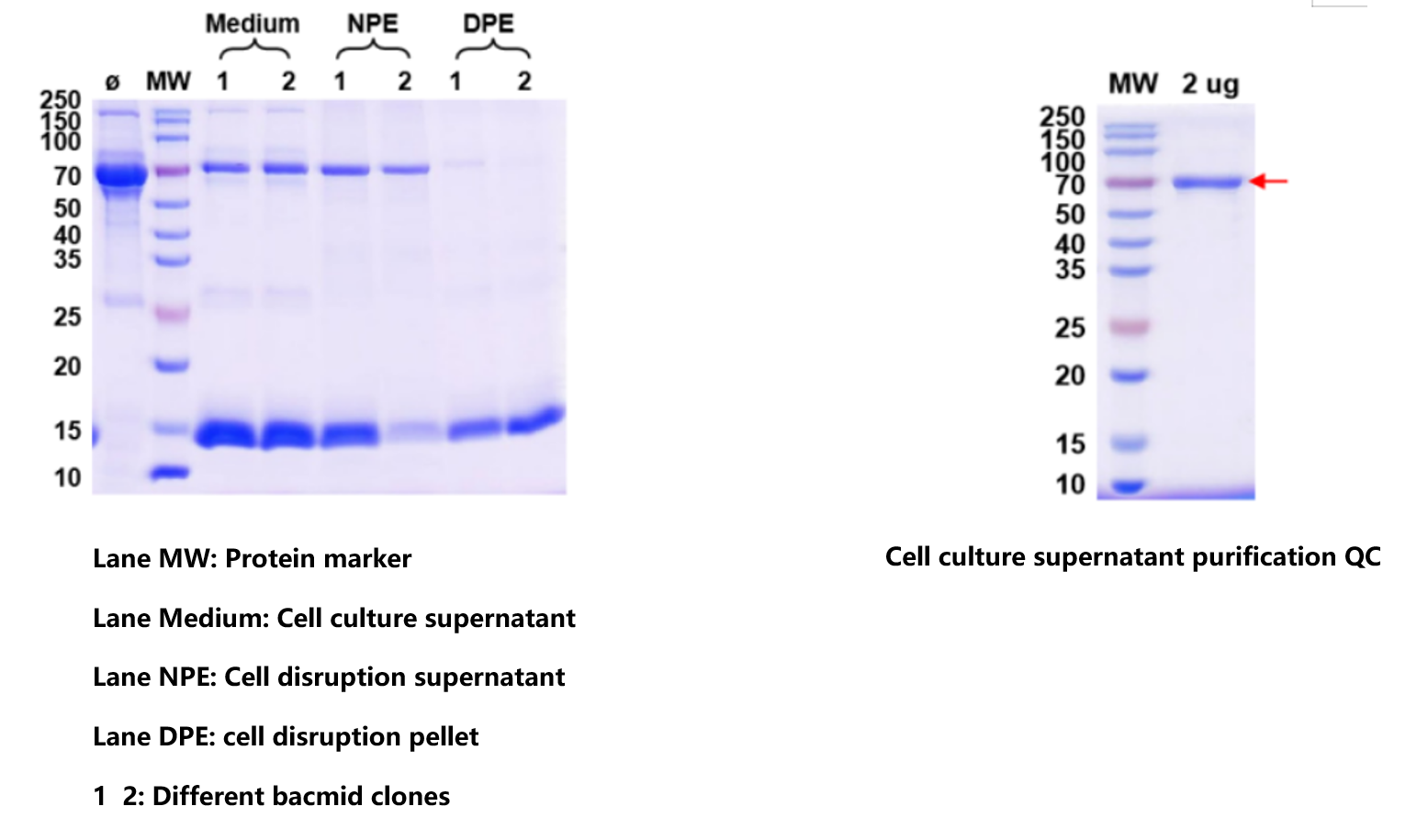
Figure 1. Comprehensive QC panel for recombinant antibody expression. Data show strong target signals with low background across purity and functional readouts, confirming robust expression yield, high purity, correct assembly, and preserved antigen-binding activity — meeting release criteria for downstream research applications.
The recombinant antibody met all release specifications: correct molecular weight on SDS-PAGE (both chains present under reducing conditions, intact IgG under non-reducing), high monomer content, and specific antigen recognition in functional assays with low background signal. The data confirm a stable, reproducible workflow capable of delivering assay-grade antibody material from sequence to validated reagent.
| Deliverable | Specification |
|---|---|
| Purified antibody | Typically 1–10 mg (scalable to gram upon request) |
| SDS-PAGE | Reducing + non-reducing; purity >90% (typically >95%) |
| SEC-HPLC | Monomer content >95%; aggregate and fragment quantification |
| Endotoxin | <1 EU/mg (LAL assay); <0.1 EU/µg available upon request |
| Functional validation | Antigen-binding ELISA standard; WB, IHC, IF, FC, SPR/BLI optional |
| Sequence | VH/VL sequences provided (FASTA + annotated); full plasmid map on request |
| COA + report | Lot-specific Certificate of Analysis with all QC data and methods summary |
Why This Matters
A recombinant antibody that looks pure on SDS-PAGE but doesn't bind its antigen is worthless. Conversely, an antibody with confirmed binding but high aggregate content will produce inconsistent results across experiments. AtaGenix's QC philosophy covers both dimensions — purity and function — because an antibody is only "good" if it performs in the client's actual assay. Every batch ships with data proving both, not just one.
This case study represents an anonymized project outcome. Results may vary depending on antibody sequence, expression system, and downstream application. All client information is subject to NDA.
Need recombinant antibody expression with guaranteed functional validation? AtaGenix delivers sequence-to-antibody with comprehensive QC — from milligram research batches to gram-scale production.
Start Your Antibody Expression ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

