AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-11-05 View volume: 999
Overview — Two case studies demonstrating AtaGenix's CHO stable cell line platform: (1) a therapeutic antibody program achieving >3 g/L titer with comprehensive QC validation, and (2) a viral protein (VZV gE) stable line for vaccine-related applications. Both projects illustrate AtaGenix's end-to-end capability from gene design through validated cell bank delivery.
A biopharma client required a clonally-derived CHO production line for a therapeutic monoclonal antibody, with the goal of achieving gram-per-liter titers in fed-batch culture while meeting stringent quality criteria for downstream CMC development. The cell line needed to demonstrate long-term expression stability, high product purity, and sub-nanomolar target binding affinity.
AtaGenix deployed its CHO-K1 stable cell line platform with the following workflow:
The top-performing clone achieved >3 g/L antibody titer in fed-batch culture, with stable expression maintained across multiple passages. The complete QC package confirmed the cell line was ready for further CMC development:
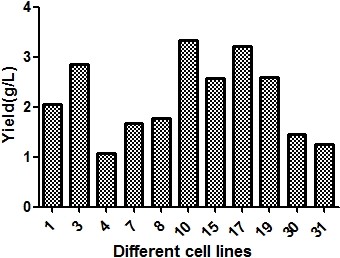
Figure 1. Antibody monoclonal yield analysis. Clone-to-clone screening revealed a wide titer range, with the top-performing clone exceeding 3 g/L in fed-batch culture — demonstrating the value of high-throughput screening for identifying rare high producers.
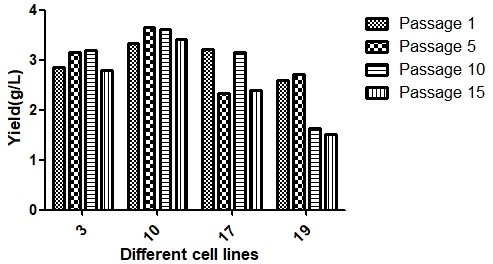
Figure 2. Expression stability across passages. Antibody titer remained consistent over extended culture, confirming long-term production reliability essential for manufacturing campaigns.
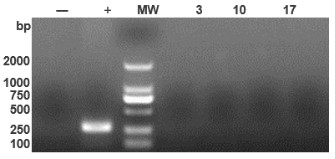
Figure 3. Mycoplasma testing (PCR method). MW: DL2000 marker; (+): positive control (290 bp); (-): negative control. All cell bank samples tested negative, confirming biosafety compliance.
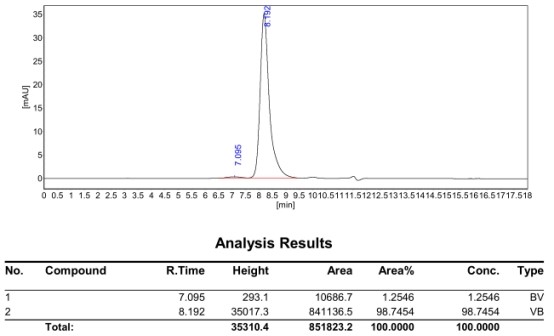
Figure 4. Antibody purity analysis (SDS-PAGE). The top clone produced antibody with high purity under both reducing and non-reducing conditions, with minimal host cell protein contamination.
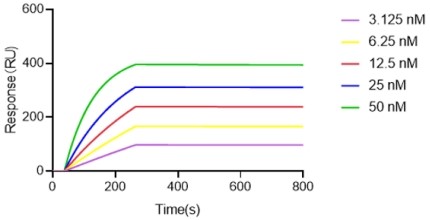
| Sample | ka (1/Ms) | kd (1/s) | KD (M) |
|---|---|---|---|
| 12-10 | 1.29E+05 | 9.52E-06 | 7.36E-11 |
Figure 5. Antibody binding kinetics (SPR). The top clone antibody demonstrated sub-nanomolar affinity (KD = 7.36 × 10-11 M), with fast association and slow dissociation rates — confirming suitability for therapeutic development.
A vaccine-focused client needed a stable CHO production line for Varicella-Zoster Virus glycoprotein E (VZV gE) — the lead antigen candidate for a recombinant shingles vaccine. Unlike transient expression which provides protein for early characterization, stable cell line production ensures the consistent, scalable antigen supply required for preclinical immunogenicity studies and eventual manufacturing.
AtaGenix applied the same CHO-K1 stable cell line platform used for antibody programs, adapted for a viral glycoprotein target. The gE ectodomain was codon-optimized for CHO expression, cloned with an appropriate signal peptide for secretion, and transfected into CHO-K1 cells under selection pressure. Multiple clones were screened and the top expressors were evaluated by SDS-PAGE for correct molecular weight, band intensity, and product homogeneity.
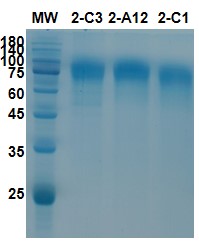
Figure 6. SDS-PAGE analysis of VZV gE protein from different CHO stable clones. MW: protein marker. Distinct bands at expected molecular weight confirm successful gE expression across multiple clones, supporting downstream clone selection for vaccine antigen production.
Multiple clones showed robust gE expression with correct molecular weight and minimal degradation products. The lead clone was advanced to cell banking with the same QC standards applied in the antibody program (stability, mycoplasma, purity), providing the client with a reliable, renewable antigen source for their vaccine development pipeline.
Why This Matters
A stable cell line is a long-term asset. Unlike transient expression that provides a one-time protein batch, a validated cell line with a cryopreserved Research Cell Bank delivers protein on demand for years — with the same quality every time. These two case studies demonstrate that AtaGenix's CHO platform is versatile enough to handle both antibodies (>3 g/L titer with picomolar affinity) and non-antibody targets (viral glycoproteins for vaccines), with a unified QC framework covering productivity, stability, purity, biosafety, and functional binding.
These case studies represent anonymized project outcomes. Results may vary depending on target protein, construct design, and project scope. All client information is subject to NDA.
Need a stable cell line for antibodies, Fc-fusions, or viral antigens? AtaGenix delivers clonally-derived CHO production lines with full QC documentation — from gene to Research Cell Bank in approximately 12 weeks.
Discuss Your Cell Line ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

