AtaGenix Laboratories
AtaGenix Laboratories
Release time: 2024-11-05 View volume: 1104
Overview — E. coli is fast and cheap, but it comes with baggage: endotoxin. For proteins headed into animal studies, diagnostic kits, or food-grade applications, that LPS contamination can be a dealbreaker. These two case studies show how AtaGenix's Bacillus subtilis WB800N platform sidesteps the endotoxin problem entirely — delivering secreted Protein A at 20 mg/L (>90% purity) and intracellular Protein B at >95% purity, without a single endotoxin removal column in the workflow.
Most researchers default to E. coli for prokaryotic expression. It's familiar, well-tooled, and delivers high yields. But E. coli produces lipopolysaccharide (LPS) endotoxin as a component of its outer membrane, and removing it adds cost, complexity, and risk of product loss. For many downstream applications — in vivo injection, cell-based assays, diagnostic manufacturing, food enzymes — endotoxin is not just inconvenient, it's a regulatory barrier.
Bacillus subtilis is a Gram-positive bacterium with no outer membrane and therefore no endotoxin. It holds GRAS (Generally Recognized As Safe) status from the FDA, grows rapidly in simple media, and naturally secretes proteins into the culture supernatant. AtaGenix uses the WB800N strain — engineered with eight extracellular protease gene knockouts — to further protect secreted products from degradation. The result: clean protein from culture supernatant, no cell lysis needed, no endotoxin removal needed.
| Feature | B. subtilis WB800N | E. coli BL21 |
|---|---|---|
| Endotoxin | None (Gram+) | Present (requires removal) |
| GRAS Status | Yes | No |
| Secretion | Efficient (native pathway) | Limited (periplasm only) |
| Protease Activity | Minimal (WB800N: 8 KOs) | Low (BL21 lon/ompT KO) |
| Typical Yield | 1–50 mg/L | 50–500 mg/L |
| Best For | In vivo-grade, food enzymes, diagnostics | High-throughput screening, structural biology |
Client (NDA) | Diagnostic Reagent Program
Challenge: The client needed a large recombinant protein (~85 kDa) as a diagnostic reagent component. The protein had to be endotoxin-free without relying on removal chromatography — their manufacturing process could not accommodate the additional step without exceeding cost targets. Previous attempts with E. coli achieved good yield but endotoxin levels remained above the <0.5 EU/mg threshold required for their immunoassay application.
Solution: AtaGenix synthesized the gene with B. subtilis codon optimization, subcloned it into pHT43 (secretion vector with signal peptide), and transformed into WB800N. IPTG-induced expression directed the N-terminal 6×His-tagged protein into the culture supernatant. Ni-NTA affinity chromatography from clarified supernatant — no cell lysis, no inclusion body processing, no endotoxin removal — yielded purified protein at 20 mg/L with >90% purity.
Result: The protein passed the client's incoming QC (<0.1 EU/mg endotoxin, >90% purity by SDS-PAGE) on the first delivery — no rework, no endotoxin polishing, no additional costs. Multiple clones were screened to provide the client with backup options for future scale-up.
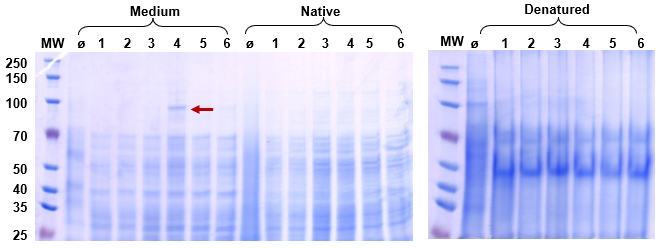
Figure 1. Reduced SDS-PAGE of Protein A secretion expression. Lane M: protein marker; Lane Ø: blank control; Lanes 1–6: supernatant and lysate fractions from different WB800N clones. Clear ~85 kDa bands in supernatant lanes confirm successful signal peptide-mediated secretion — the protein was purified directly from culture medium without cell lysis.
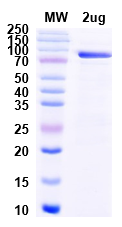
Figure 2. Extended QC analysis of purified Protein A: identity confirmation, purity assessment, and concentration measurement. All specifications met on first production run.
Client (NDA) | Research Reagent Program
Challenge: A smaller protein (~42 kDa) was needed at the highest possible purity for structural characterization. However, pilot secretion tests showed the protein was rapidly degraded in the extracellular environment — even in the protease-reduced WB800N background. The client still needed endotoxin-free production, ruling out E. coli.
Solution: AtaGenix switched to pHT254, an intracellular expression vector that retains the C-terminal 6×His-tagged protein inside the cytoplasm, protecting it from extracellular proteases. After IPTG induction, cells were lysed under controlled conditions and soluble protein was recovered from the lysate supernatant. Ni-NTA affinity chromatography yielded 1 mg/L at >95% purity — lower volumetric yield than the secreted case (typical for intracellular expression), but at higher purity and with intact full-length protein confirmed by SDS-PAGE.
Result: The >95% purity met the client's threshold for downstream biophysical characterization. Critically, the protein remained endotoxin-free despite requiring cell lysis — because the host organism itself produces no LPS, there is nothing to contaminate the lysate.
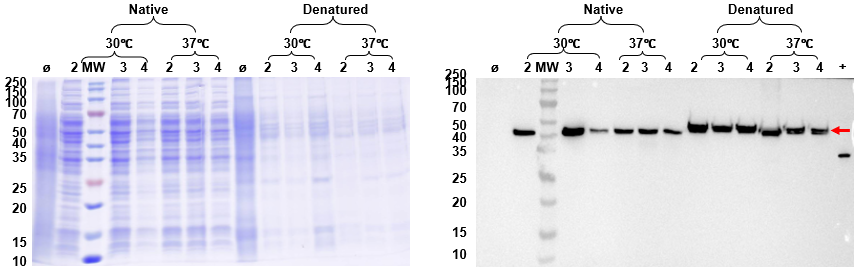
Figure 3. Reduced SDS-PAGE of Protein B intracellular expression. Lane M: protein marker; Lane Ø: blank control; Lane +: positive control; Lanes 2–4: lysate supernatant and precipitate from different clones. Target band at ~42 kDa confirmed in the soluble fraction, with minimal degradation products.
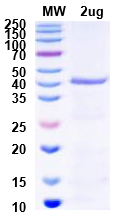
Figure 4. Extended QC analysis of purified Protein B. >95% purity confirmed, meeting specifications for structural characterization and biophysical studies.
| Secreted (pHT43) | Intracellular (pHT254) | |
|---|---|---|
| Purification | From supernatant (no lysis) | From lysate (cell disruption) |
| Typical Purity | >90% | >95% |
| Protease Risk | Minimal (WB800N 8-KO) | Very low (cytoplasmic) |
| Best For | Stable proteins, simplified workflow | Degradation-prone targets, max purity |
| Endotoxin | Free | Free |
Why This Matters
Endotoxin removal from E. coli-produced proteins typically adds 1–2 days of processing time, reduces yield by 20–40%, and still risks residual LPS if the removal is incomplete. For applications where endotoxin is a hard specification — in vivo studies, cell-based assays, diagnostic manufacturing — starting with an LPS-free host eliminates the problem at the source. These case studies show that B. subtilis isn't just a niche alternative; it's a rational first choice when endotoxin-free production matters, and AtaGenix offers both secreted and intracellular strategies to match your protein's properties.
These case studies represent anonymized project outcomes. Results may vary depending on target protein, construct design, and project scope. All client information is subject to NDA.
Need endotoxin-free protein production without the hassle of LPS removal? AtaGenix offers B. subtilis expression alongside E. coli, yeast, insect, and mammalian platforms — our scientists will recommend the best system for your target and application.
Discuss Your Expression ProjectResponse within 24 hours
Contact Us
+86-27-6552-3339
info@atagenix.com
Building C, R & D Building, No. 666, Shendun 4th Road, Donghu New Technology Development Zone, Wuhan

